
Concept explainers
How would each of the following pairs of compounds differ in their IR spectra?
a. 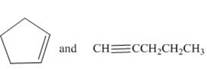 d.
d.![]()
b.![]() e.
e.
c.![]() f.
f.
(a)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR spectrum of cyclopentene is different from pentyne because the absorption of
Explanation of Solution
The given compounds are cyclopentene and pentyne as shown below.
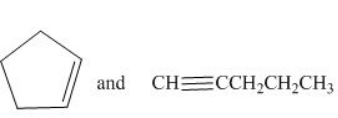
Figure 1
Cyclopentene contains
The IR spectrum of cyclopentene is different from pentyne because the absorption of
(b)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR absorption of propanoic acid is different from methyl acetate due to broad peak of
Explanation of Solution
The given compounds are propanoic acid and methyl acetate as shown below.
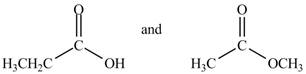
Figure 2
Propanoic acid contains
The IR absorptions of propanoic acid is different from methyl acetate due to broad peak of
(c)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR absorptions of butan-
Explanation of Solution
The given compounds are butan-

Figure 3
Butan-
The IR absorptions of propanoic acid is different from methyl acetate due to broad peak of
(d)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR spectra of methyl hexanoate are different from
Explanation of Solution
The given compound are
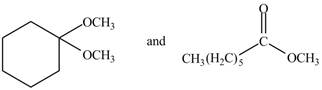
Figure 4
Methyl hexanoate contains
The IR spectra of methylhexanoate is different from
(e)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
But-
Explanation of Solution
But-
But-
(f)
Interpretation: The given pair of compounds is to be differentiated on the basis of their IR spectra.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.40P
The IR spectra of N, N-diethylprop-
Explanation of Solution
The given compound are N, N-diethylprop-
The IR spectra of N, N-diethylprop-
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry -Study Guide / Solution Manual (Custom)
- a) what bond is present in ketones but not alkanes? b) what is the wavenumber range of the signals that are present in the diagnostic region of all of the ketone IR spectra (but not the alkanes)? c) is the new band strong or weak compared with a C=C or a C≡C stretch?arrow_forwardHere are proton NMR data for 1-bromopropane: Ha : triplet (2H) 3.32ppm; Hb : multiplet (2H)1.81ppm; Hc : triplet (3H) 0.93ppm. (Relative integrations shown in parentheses.) a. Through how many bonds can a hydrogen split another hydrogen? b. According to this splitting rule, does Ha split Hc ? c. Is your answer in part a) consistent with the multiplicity listed for peak clusters a and c? d. How many hydrogenssplit Hb ? e. Upon very close inspection of the proton NMR spectrum of 1-bromopropane, you wouldfind that peak cluster b has at least six peaks. Is this consistent with your answer in part d)? f. Speculate as to why any peak cluster with more than four peaks is listed simply as a"multiplet."arrow_forwardFollowing are infrared spectra of nonane and 1-hexanol. Assign each compound its correct spectrum.arrow_forward
- Would you expect two enantiomers such as (R)-2-bromobutane and (S)-2-bromobutane to have identical or different IR spectra? Explain.arrow_forwardFor each structure below, use numbers to indicate chemically equivalent and distinct hydrogens,and make a table showing the predicted integration and multiplicity of each peak cluster.arrow_forwardFollowing are infrared spectra of 2-methyl-1-butanol and tert-butyl methyl ether. Assign each compound its correct spectrum.arrow_forward
- Following are mass spectra for the constitutional isomers 2-pentanol and 2-methyl- 2-butanol. Assign each isomer its correct spectrum.arrow_forwardThe hormone cortisone contains C, H, and O, and shows a molecular ion at M+=360.1937 by high-resolution mass spectrometry. What is the molecular formula of cortisone? (The degree of unsaturation for cortisone is 8.)arrow_forwarda) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of spectra)? c) what is different about C=O and O-H bonds compared with C=C or C≡C bonds that show weaker signals?arrow_forward
- How could IR spectroscopy distinguish between 1,5-hexadiene and 2,4-hexadiene?arrow_forwardTHIS CAN NOT BE HAND - DRAWN Illustrate using a computer program the molecule from the molecular formula and interpret each of the following 1H NMR spectra. The integration for each peak is given above the peak. All spectra were taken from http://riodb01.ibase.aist.go.jp/sdbs/cgi-bin/direct_frame_top.cgi.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



