
ORGANIC CHEMISTRY LL BUNDLE
4th Edition
ISBN: 9781119761112
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 13, Problem 52ASP
Interpretation Introduction
Interpretation:
The best retrosynthesis of the given target molecule needs to be identified.
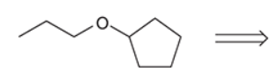
Concept Introduction: The preparation of ether takes place by the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write a reaction sequence of 4 steps and, afterwards, write the retrosynthesis
propose a multi-step pathway for the desired target compound.
Here is the same figure as in the previous two questions, but omitting the red circles.
Cys157
Cys157
Cys157
His296
His296
His296
LOH
HN
HN
HN N?
-CO2
CH2
Asn329
Asn329
Asn329-
Cys157
His296
LOH
HN H HyC.
Asn329
Which of the following roles is played by the imidazole side chain of His296? Pick the one best
[ Select ]
answer.
Chapter 13 Solutions
ORGANIC CHEMISTRY LL BUNDLE
Ch. 13.2 - Prob. 1LTSCh. 13.2 - Prob. 1PTSCh. 13.2 - Prob. 2PTSCh. 13.2 - Prob. 3ATSCh. 13.4 - Prob. 4CCCh. 13.5 - Prob. 2LTSCh. 13.5 - Prob. 5PTSCh. 13.5 - Prob. 6ATSCh. 13.5 - Prob. 7CCCh. 13.5 - Prob. 8CC
Ch. 13.5 - Prob. 9CCCh. 13.6 - Prob. 10CCCh. 13.7 - Prob. 11CCCh. 13.7 - Prob. 12CCCh. 13.8 - Prob. 3LTSCh. 13.8 - Prob. 13PTSCh. 13.8 - Prob. 14ATSCh. 13.9 - Prob. 15CCCh. 13.10 - Prob. 4LTSCh. 13.10 - Prob. 17ATSCh. 13.10 - Prob. 5LTSCh. 13.10 - Prob. 19ATSCh. 13.11 - Prob. 20CCCh. 13.12 - Prob. 6LTSCh. 13.12 - Prob. 7LTSCh. 13 - Prob. 26PPCh. 13 - Prob. 27PPCh. 13 - Prob. 28PPCh. 13 - Prob. 29PPCh. 13 - Prob. 30PPCh. 13 - Prob. 31PPCh. 13 - Prob. 32PPCh. 13 - Prob. 33PPCh. 13 - Prob. 34PPCh. 13 - Prob. 35PPCh. 13 - Prob. 36PPCh. 13 - Prob. 37PPCh. 13 - Prob. 38PPCh. 13 - Prob. 39PPCh. 13 - Prob. 40PPCh. 13 - Prob. 41PPCh. 13 - Prob. 42PPCh. 13 - Prob. 43PPCh. 13 - Prob. 44PPCh. 13 - Prob. 45PPCh. 13 - Prob. 46ASPCh. 13 - Prob. 47ASPCh. 13 - Prob. 48ASPCh. 13 - Prob. 49ASPCh. 13 - Prob. 50ASPCh. 13 - Prob. 51ASPCh. 13 - Prob. 52ASPCh. 13 - Prob. 53ASPCh. 13 - Prob. 54IPCh. 13 - Prob. 59IPCh. 13 - Prob. 60IPCh. 13 - Prob. 61IPCh. 13 - Prob. 62IPCh. 13 - Prob. 63IPCh. 13 - Prob. 64IPCh. 13 - Prob. 65IPCh. 13 - Prob. 66IPCh. 13 - Prob. 69IPCh. 13 - Prob. 70IPCh. 13 - Prob. 71IPCh. 13 - Prob. 72IPCh. 13 - Prob. 73IPCh. 13 - Prob. 74IPCh. 13 - Prob. 77CPCh. 13 - Prob. 79CPCh. 13 - Prob. 80CP
Knowledge Booster
Similar questions
- what are the limitations of Williamson's synthesis?arrow_forward1. Which of the following statement about safety is NOT CORRECT? A) Approved safety goggles must be worn at all times you are working in the laboratory. B) No food, drinks or smoking are allowed in the laboratory. C) Wear gloves when you are dealing with chemicals. D) Broken or cracked glassware can be used in the experiments. E) No open-toe shoes are allowed in the laboratory. 5. Which of the following is an Aldol condensation? A) Two molecules of benzaldehyde react with each other to form an alpha-hydroxy ketone B) Methanol reacts with benzoic acid to form methyl benzoate. C) Aldehydes react with methyl ketones to form alpha, beta-unsaturated ketones. D) Nitration of methyl benzoate to form methyl 3-nitrobenzoate. E) Acylation of ferrocene to form acetylferrocene. 4. Which of the following is(are) the minor product(s) in the nitration of nitrobenzene? A) Nitrobenzene B) 1,2-Dinitrobenzene C) 1,3-Dinitrobenzene D) 1,4-Dinitrobenzene E) 1,2-Dinitrobenzene and 1,4-dinitrobenzene 3.…arrow_forwardWhat is the reagent R1 in this reaction sequence?arrow_forward
- Use E1, sn1 or E2, sn2 Synthesisarrow_forwardTwo methods convert an alkyl halide to a carboxylic acid having one more carbon atom. Depending on the structure of the alkyl halide, one or both of these methods may be employed. For each alkyl halide, write out a stepwise sequence that converts it to a carboxylic acid with one more carbon atom. If both methods work, draw both routes. If one method cannot be used, state why it can't.arrow_forwardWhat are the compounds X,Y and Z in the given reaction sequence ?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning