
a.
Interpretation:
Product that forms has to be drawn when the given amide is treated with water and sulfuric acid.
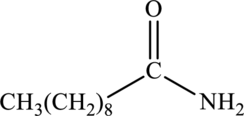
Concept Introduction:
Amide also undergo hydrolysis as like esters. They are much less reactive than the esters. When amide is treated with water in presence of acid as a catalyst, the product formed is carboxylic acid and ammonium salt. General reaction can be given as follows.
b.
Interpretation:
Product that forms has to be drawn when the given amide is treated with water and sulfuric acid.
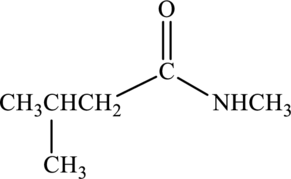
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
Product that forms has to be drawn when the given amide is treated with water and sulfuric acid.
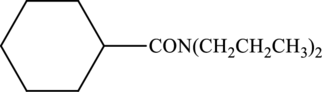
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- What 1° amine and carbonyl compound are needed to prepare each imine?arrow_forwardWhat nitro compound, nitrile, and amide are reduced to each compound?arrow_forward18-46 Procaine (its hydrochloride salt is marketed as Novocaine) was one of the first local anesthetics developed for infiltration and regional anesthesia. it is synthesized by the following fischer esterification:arrow_forward
- What products are formed by hydrolysis of each imine or enamine?arrow_forwardDraw the structural formula for each amides. - N-Cycloclohexylacetamide - Benzamidearrow_forwardWhat products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forward
- What product is formed when each carbonyl compound is treated with K2Cr2O7?arrow_forwardDraw the products formed when the given amides are treated with aqueous potassium hydroxide. Please draw the products as skeletal structures. Thanks!arrow_forwardWhat carbonyl compound and amine or alcohol are needed to prepare each product?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

