
Concept explainers
a.
Interpretation:
The ester that is formed when the given
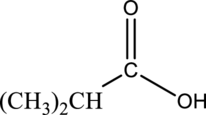
Concept Introduction:
In presence of acid, the carboxylic acid and an alcohol reacts to form ester as product. Acid acts as a catalyst in this reaction. This is known as Fischer esterification reaction. Esterification reaction is an example of substitution reaction because, the
b.
Interpretation:
The ester that is formed when the given carboxylic acid reacts with ethanol in presence of sulfuric acid has to be drawn.
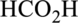
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
The ester that is formed when the given carboxylic acid reacts with ethanol in presence of sulfuric acid has to be drawn.
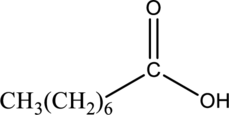
Concept Introduction:
Refer part “a.”.
d.
Interpretation:
The ester that is formed when the given carboxylic acid reacts with ethanol in presence of sulfuric acid has to be drawn.
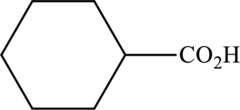
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the carbonyl products formed when each alcohol is oxidized with K 2Cr 2O 7.arrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forwardWhat alcohol is formed when each compound is treated with NaBH4 in CH3OH?arrow_forward
- What are the products formed when 1-butanal reacts with: NaOH/H2O CrO3, H2SO4, H2Oarrow_forwardWhat products are formed when benzene is treated with each alkyl chloride and AlCl3?arrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. c. CH3CH2Cl, AlCl3 l. product in (c), then KMnO4arrow_forward
- Draw the product formed when the alcohol cyclobutanol is dehydrated with H2SO4.arrow_forwardWhat products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forwardDraw the hemiacetal and acetal formed when the carbonyl compound is treated with two equivalents of the given alcohol in the presence of H2SO4.arrow_forward
- Benzaldehyde is the compound principally responsible for the odor of almonds. What product is formed when benzaldehyde is treated with H2, Pd?arrow_forwardRank the alcohols in order of increasing reactivity when dehydrated with H2SO4.arrow_forwardWhat product is formed when each alkene is treated with HCl?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





