
Concept explainers
a.
Interpretation:
Amine given has to be named.
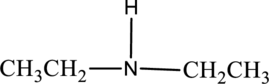
Concept Introduction:
- In case of primary amine, to the nitrogen atom, one alkyl group is bonded. The name of the alkyl group is written followed by suffix –amine resulting in a single word.
- In case of secondary and tertiary amine, the different alkyl groups are named as in case of primary amine but arranged in alphabetical order. If there are identical alkyl groups attached to nitrogen atom, then suitable prefix di- or tri- is used.
b.
Interpretation:
Acceptable name has to be given for the below compound.
Concept Introduction:
Amides contain a nitrogen atom that is bonded to carbonyl group. The nitrogen atom that is bonded to the carbonyl group may be bonded to alkyl groups or hydrogen atoms. Depending on the number of carbon atoms that is attached to the nitrogen atom, the amide is classified as primary, secondary, and tertiary.
Naming of amides are done using specific rules:
Primary amides are named by considering the parent carboxylic acid and replacing the “–oic acid” with “–amide”.
Secondary and tertiary amide contains two parts in its name. The alkyl groups that are bonded to the nitrogen atom is named using N- as the prefix for the alkyl group and this precedes the name. The
b.
Interpretation:
Acceptable name has to be given for the below compound.
Concept Introduction:
Refer part “b.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- 1. What are the hazards and benefits of Amines? 2. What are the hazards and benefits of Nitriles? 3. What are the hazards and benefits of Nitro Compounds?arrow_forwardMatch the description to one of the compounds E– H. a. a compound that contains a 1 ° amine and a 1 ° amide b. a compound that contains a 1 ° amine and a 2 ° amide c. a compound that contains a 2 ° amine and a 3 ° amide d. a compound that contains a 3 ° amine and a 3 ° amidearrow_forwardWhich type of amine is phentermine? a) a primary aliphatic amine b) a primary aromatic amine c) a tertiary aliphatic amine d) a tertiary aromatic aminearrow_forward
- Draw a structural formula for each amine and amine derivative. Q.) trans-2-Aminocyclohexanolarrow_forwardN-p-hydroxyphenylethanamide is commonly known as a. acetaminophen b. acetamide c. acetanilide d. formamide High molar mass amines have __________ odor. a.strong ammoniacal b.fruity c.fishy d.obnoxious Trimethyl amine has _________ odor. a.obnoxious b.fishy c. ammoniacal d. fruityarrow_forwardA) Name the following amine. H3C−CH2−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound. B ) Name the following amine. CH3−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound.arrow_forward
- Which of the following is an amine? Select one: a. HCONH2 b. CH3COCH3 c. CH3F d. CH3NH2arrow_forwardLabel each nitrogen-containing functional group in lidocaine, a local anesthetic, as an amine or amide, and classify it as 1 °, 2 °, or 3 °.arrow_forwardWhat ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forward
- Draw a structural formula for each amine and amine derivative. Q.) N,N-Dimethylanilinearrow_forwardWhich type of amine is (s)-methamphetamine? a) a primary aliphatic amine b) a primary aromatic amine c) a secondary aliphatic amine d) a secondary aromatic aminearrow_forwardDraw a structural formula for each amine and amine derivative. Q.) Benzylaminearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



