
a.
Interpretation:
Amide given below has to be named.
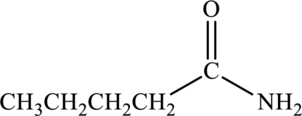
Concept Introduction:
Amides contain a nitrogen atom that is bonded to carbonyl group. The nitrogen atom that is bonded to the carbonyl group may be bonded to alkyl groups or hydrogen atoms. Depending on the number of carbon atoms that is attached to the nitrogen atom, the amide is classified as primary, secondary, and tertiary.
Naming of amides are done using specific rules:
Primary amides are named by considering the parent carboxylic acid and replacing the “–oic acid” with “–amide”.
Secondary and tertiary amide contains two parts in its name. The alkyl groups that are bonded to the nitrogen atom is named using N- as the prefix for the alkyl group and this precedes the name. The
b.
Interpretation:
Amide given below has to be named.
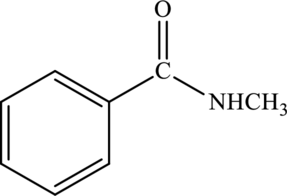
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
Amide given below has to be named.
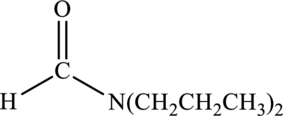
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- draw structure for: 2-formylethanamidearrow_forwardWhat are the functional groups present in this antibacterial antibiotic? A. Amide, thioether, aldehyde, phenol, carboxylic acid B. Amide, thioether, ketone, amine, phenol, carboxylic acid C. Amide, thioether, ketone, phenol, carboxylic acid D. Thioether, ketone, amine, phenol, carboxylic acid A brief explanation would be highly appreciated + upvotearrow_forwardIdentify the characteristics of aminesarrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



