
Concept explainers
a.
Interpretation:
The compound
Concept Introduction:
Ammonium salts are similar to ionic compounds. They have cation and anion. Ammonium salts are highly soluble in water.
b.
Interpretation:
The compound
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
The compound
Concept Introduction:
Refer part “a.”.
d.
Interpretation:
The given compound is water soluble or water insoluble has to be labelled.
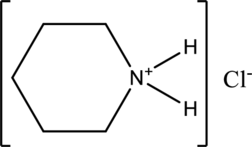
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
- Explain why m-nitroaniline is a stronger base than p-nitroaniline.arrow_forwardComplete each acid-base reaction and predict whether the position of equilibrium lies toward the left or toward the right. (a) CH3CCH+CH3CH2ONa+CH3CH3OH (b) CH3CCCH2CH2OH+Na+NH2NH3(l)arrow_forwardWhich is a stronger base? a. CH3O− or CH3S− b. H2O or HO− c. H2O or NH3 d. CH3CO− or CH3O−arrow_forward
- Draw the products when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents no reaction occurs: a)NH3,∆ B)1) SOCl, 2) CH3CH2CH2NH2 (excess) C) 1)SOCl2, 2) (CH3)2CHOHarrow_forwardK15. rank from weakest to strongest Bronsted base. Please give explanation and what the rankings look like for this questionarrow_forwardWhat reagent can be used from compound F to G? (NaH / NaOH / LiAlH4+hydronium quench / CrO3 Jones)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


