
Concept explainers
Interpretation:
The reason should be explained for glucose is more soluble than 1-hexanol.
Concept introduction:
Solubility of the alcohol in water:
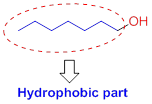
The solubility of the alcohol is mainly depends on the hydrophilic part as well as length of the alkyl chain. If the compound has more number of hydroxyl group or less number of carbon chain which is soluble in water. If the compound has less number of hydroxyl group with more number of carbon chain which is insoluble in water because of hydrophobic part.
Alcohols have two different parts
- (1) Hydrophilic (water-loving),
- (2) Hydrophobic (water-fearing).
Hydrophilic (water-loving):
The compound has the hydroxyl group (

Hydrophobic (water-fearing):
The compound has alkyl chain group is called as hydrophilic.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Study Guide And Full Solutions Manual For Fundamentals Of General, Organic, And Biological Chemistry
- D-Erythrose and D-threose are referred to as _____________ because their structures only differ in the configuration at one asymmetric carbon atom.arrow_forwardTrehalose, a disaccharide found in the blood of insects, has the following structure. What simple sugars would you obtain on hydrolysis of trehalose?arrow_forwardThis is the structure of 2,4-dimethyl-1-pentanol.arrow_forward
- Glucose-1-phosphate has a ΔG°′ value of −20.9 kJ/mol, whereas that for glucose-6-phosphate is −12.5 kJ/mol. After reviewing the molecular structures of these compounds, explain why there is such a difference in these values.arrow_forwardWhy is every uracil replaced by 1-methyl-3'-pseudouridylyl? Explain why it is included please.arrow_forwardThe ring structure of the carbohydrate glucose is provided below. Map any chiral centers with a 1.arrow_forward
- What structural relationship is indicated by term D-sugar? Why are +glucose and -fructose classified as D-sugarsarrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forwardWhat is unusual about glutathione’s structure? (If you cannot answer this question, draw the structure you would expect for the tripeptide and compare your structure with the actual structure.)arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





