
Concept explainers
Interpretation:
The
Concept introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid.
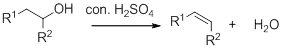
Alcohol is reaction with concentrated sulfuric acid, first alcohol gets protonated forms carbocation (more stable carbocation) followed by elimination of proton (
Tertiary carbocation is more stable than the secondary, secondary carbocation is more stable than primary.

In dehydration reaction, sulfuric acid is act as a proton donor, and which is used to protonate the alcohol and makes carbocation therefore sulfuric acid is the driving force of the reaction. Dehydration reaction will not go without acid (sulfuric acid).
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
- If the dehydration reaction of an alcohol is successful, what changes would be seen in the IR spectrum for the product compared to the starting materialarrow_forwardGive the reagent or chemical test that would differentiate the following pairs of compounds. Write chemical equations for the reactions. List of Chemical Tests (choose here)I. Halogenation: Bromination with and without lightII. Oxidation: Reaction with Baeyer’s reagent III. Reaction with Ammoniacal Silver Nitrate (AgNO3) or simply Tollen’s reagent a. benzene and ethylbenzeneb. 1-butyne and 2-butynec. 2-methylpentane and 2-methyl-2-pentened. toluene and 1-methylcyclohexenearrow_forwardA prescription came in for 34% salicylic acid. The pharmacy has 95% salicylic and 10%salicylic acid available. Write a recipe for 500mg of 34% salicylic acid.arrow_forward
- What functional group is formed on oxidation of a secondary alcohol? Demonstrate your answer using isopropanol.arrow_forwardThe concentration of alcohol 1CH3CH2OH2 in blood, calledthe “blood alcohol concentration” or BAC, is given in unitsof grams of alcohol per 100 mL of blood. The legal definitionof intoxication, in many states of the United States, is that theBAC is 0.08 or higher. What is the concentration of alcohol,in terms of molarity, in blood if the BAC is 0.08?arrow_forward1. Rank the following amines from lowest to highest boiling point. Explain your reasoning. CH3 CH2CH3 H3C CH3 ༣.མིའི་ན་བྱིན་ CH NH₂ -CH3 H₂ NH2 2. Which compound in each pair would have the higher boiling point? Why? a. H3C CH3 H3C OH b. NH2 CH3 H₂ H3C CH CH CH3 H3C -CH3 NH2 3. Circle each of the following molecules that would be significantly soluble in water. Explain. ΝΗΣ H₂ H₂ H₂ H₂ CH C- NH₂ H3C -CH3 H3C H2 H₂ CH3 CH CH3 H3C -CH3 H3C H₂arrow_forward
- Compare the densities of low-density polyethylene (LDPE) and high-density poly- ethylene (HDPE) with the densities of the liquid alkanes.How might you account for the differences between them?arrow_forwardThe reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardA 5 ml vial of hydrocortisone is having 2% (w/v) concentration. How many vials will be required, if we need to prepare 30 ml of a 8 mg/ml oral hydrocortisone?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





