
(a)
Interpretation:
The chiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.
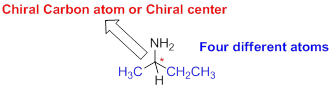
Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(b)
Interpretation:
The chiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(c)
Interpretation:
The chiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(d)
Interpretation:
The chiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(e)
Interpretation:
The chiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(f)
Interpretation:
The chiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
- Draw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardDrawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forwardWhat functional groups are present in the peaks?arrow_forward
- Each of the following materials has an ester that is responsible for its smell and/or flavor. Search the internet and determine what that ester is, draw its structure, and what carboxylic acid and alcohol are used to form it.(a) Juicy Fruit gum flavoring(b) Peach odor(c) Apple odor(d) Rum odorarrow_forwardWrite the IUPAC name for each unsaturated hydrocarbon. (a) CH,=CH(CH,),CH3 CH3 CH3 H,C- (b) CH3 (c) CH3 alaTTarrow_forwardCommon names for some triacylglycerols depend on their source. Identify the source. Choices are plant oils (soybean, canola, corn, sunflower, and so on), beef fat, and pork fat. (a) Tallow (b) Cooking oil (c) Lardarrow_forward
- Dimethyl ether has the same molecular formula as ethanol (Problem 4.57) but very different properties. Propose a structure for dimethyl ether in which the oxygen is bonded to two carbonsarrow_forwardDraw a Fischer projection formula for the enantiomer of each of the following monosaccharides. (a to d)arrow_forwardCarvone exists as a pair of enantiomers. (R)-(-)-carvone smells like spearment, whereas (S)-(+)-carvone smells like caraway. Why do these enantiomers have different smells (i.e., different biological activity) ?arrow_forward
- Salicylic acid (o-hydroxybenzoic acid) is used as starting material to prepare aspirin. Draw the structure of salicylic acid.arrow_forwardWhen pentane is exposed to Br2 in the presence of light, a halogenation reaction occurs. Write the formulas of:(a) All possible products containing only one bromine(b) All possible products containing two bromines that are not on the same carbonarrow_forwardThe following compound can be described as a(an)arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





