
(a)
Interpretation:
The condensed or skeletal structure of the following alcohol A should be drawn:
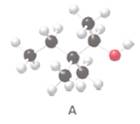
Concept Introduction:
Alcohols are the organic molecules which have OH group bonded to a tetrahedral carbon atom.
Structure of a particular molecule is the atoms and molecular arrangements through
(b)
Interpretation:
The hydroxyl group in structure A should be classified as
Concept Introduction:
Organic compounds which contains a hydroxyl group (OH) bonded to a tetrahedral carbon is known as alcohols. Alcohols are classified as
(c)
Interpretation:
The IUPAC name of the compound A should be determined.
Concept Introduction:
Alcohols are the organic molecules which have OH group bonded to a tetrahedral carbon atom.
Longest carbon chain containing the carbon bonded to the OH group is named as an
Numbering of main carbon chain is done in such a way so that OH group gets the lowest number.
When OH group is bonded to a ring, the ring is numbered beginning with the OH group and the 1 is normally omitted from the name. The ring is numbered in clockwise or anticlockwise by giving the lowest number to the next substitute.
Compounds which contains two OH groups are named as
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Glycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forwardWhich of the following is not a physical property of alcohols or phenols? A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Due to hydrogen bonding, boiling points of alcohols is much higher than those of corresponding alkanes. C. The hydroxyl group of an alcohol is nonpolar. D. Phenols are generally only slightly soluble in water.arrow_forwardWhat is the IUPAC name of the structure? a. ethanoic acid b. butanoic acid c. methyl propanoate d. ethyl propanoatearrow_forward
- 1. Carboxylic acid reacts with an alcohol to form: A. Ester and Water B. Ester C. Water D. No reaction 2. The general formula for Carboxylic acids: A. RCOOH B. RCOOR C. RCOR D. RCOH 3. General formula of phenols: * A. ROH B. Ar-OH C. R-SH D. RCOHarrow_forwardPlease explain the chosen letter. Which is NOT a physical property of alcohols or phenols? a. Phenols are generally only slightly soluble in water. b. The solubilities of normal primary alcohols in water decrease with increasing molecular weight. c. The hydroxyl group of an alcohol is nonpolar. d. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes.arrow_forward1. Draw the following compounds a. 2-methyl butanoic acid b. 3-ethyl-2-hexanonearrow_forward
- Identify the IUPAC name of the given structure. A. 2 - methylhexan-5-one B. 5 - methylhexan-2-one C. 2 - heptanone D. 5 - heptanone Identify the IUPAC name of the given structure. A. 4 - bromopentan-3-one B. 1 - bromobutan-2-one C. 2 - bromobutan-one D. None of the abovearrow_forward24 Which of the following contains a hydroxyl group? A. Alcohol B. Ether C. Thiol D. More than one answer is correct 25 Tetrahydrocannabinol molecule has four functional groups. Which one is not included? A. An alkane B. an ether C. a phenolic -OH D. benzene ring E. a carbon-carbon double bond H H3C CH3 H3C H OH CH3arrow_forward1. what group does the ff organic compound belong? a. organic nitrite b. esters c. amines d. nitriles 2. what group does the ff organic compound belong? a. alcohol b. ketone c. aldehyde d.alkene 3. what group does the ff organic compound belong? a. carboxylic acid b. alcohol c. aromatic d. ketonearrow_forward
- Which of the following is not a physical property of alcohols or phenols? Select one: A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Phenols are generally only slightly soluble in water. C. Due to hydrogen bonding, boiling points of alcohols are much higher than those of corresponding alkanes. D. The hydroxyl group of an alcohol is nonpolar.arrow_forwardB. Draw the condensed structural formula of the following alcohols. 1. 5-Ethyl-4-methyl-1-heptanol 2. 2-Methylcyclopropanol 3. 2-Mthyl-4-phenylpentan-2-olarrow_forward2. Draw the correct structure for each: a. 4-ethyl-5-isopropyl-3-methyloctane b. 6,6-diethyl-3,5,5-trimethylnonanearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





