
(a)
Interpretation:
The alcohol from which the following dehydration product can be obtained needs to be determined.
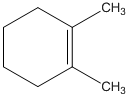
Concept Introduction:
According to Zaitsev's rule, in elimination reaction the
Dehydration reaction is defined as removal of water molecule from a compound. An alcohol reacts with acid to from alkene as a dehydration product.
(b)
Interpretation:
The alcohol from which the following dehydration product can be obtained needs to be determined.
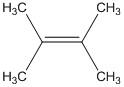
Concept Introduction:
According to Zaitsev's rule, in elimination reaction the alkene formed in major amount is the one which is formed due to the removal of hydrogen from the beta carbon atom with a smaller number of hydrogen atoms. Also, the alkene formed as major product is more substituted.
Dehydration reaction is defined as removal of water molecule from a compound. An alcohol reacts with acid to from alkene as a dehydration product.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- What is the oxidizing agent that can oxidized both primary and secondary alcohol to form aldehydes and ketones respectively?arrow_forwardWhat organic compound can be produced from the oxidation of alcohols and aldehydes? alcohols carboxylic acids ethers esters What organic reaction is displayed in the picture below? * H H NaOH H C- H Ethanol heat H H Cl H- -H Alkyl halide substitution elimination oxidation addition alkenearrow_forwardDefine Preparation of Alcohols ?arrow_forward
- What products are formed during the halogenation of an alkane?arrow_forwardExplain the characteristic reaction of aldehydes and ketones ?arrow_forward5. Dehydration of glycerin when heated in the presence of potassium hydrogen sulfate leads to the formation of:A) Unsaturated aldehydeB) Unsaturated hydrocarbonB) Unsaturated aldehyde alcoholD) Unsaturated esterarrow_forward
- Dehydration chemical reaction of alcohols in the presence of dehydrating agent can produce alkene compounds. O True O Falsearrow_forward1. What are the properties of aldehydes and ketones? 2. What will happen to aldehyde and ketones once reduced? Oxidized?arrow_forwardDraw the structure(s) of the major organic product(s) of the following reaction. aqueous H,SO. KCNarrow_forward
- Explain the reaction of aldehydes and ketones with nitrogen ?arrow_forwardAn aldehyde can react with a(an) to produce a hemiacetal. O ketone acid alcohol O alkanearrow_forwardIn the chemical reaction of reduction, which of the following is the solvent of acetone? butanol O propanol ethanol O methanol What yield when alcohols are subjected to oxidation? * ketones and aldehydes alcohols and oxygen O primary alcohols and aldehydes O aldehydes and carboxylic acidsarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning



