
Chemistry & Chemical Reactivity
9th Edition
ISBN: 9781133949640
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 68GQ
Hundreds of different reactions occur in the stratosphere, among them reactions that destroy the Earth’s ozone layer. The table below lists several (second-order) reactions of Cl atoms with ozone and organic compounds; each is given with its rate constant.
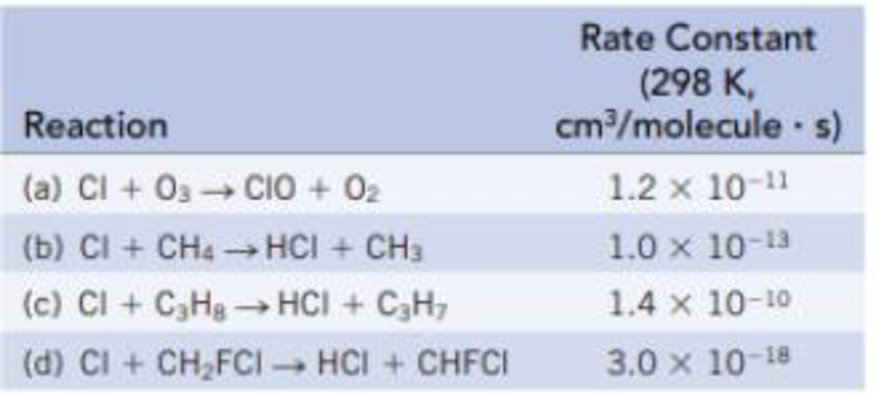
For equal concentrations of Cl and the other reactant, which is the slowest reaction? Which is the fastest reaction?
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 14 Solutions
Chemistry & Chemical Reactivity
Ch. 14.1 - Sucrose decomposes to fructose and glucose in acid...Ch. 14.1 - What are the relative rates of appearance or...Ch. 14.1 - Prob. 1RCCh. 14.1 - 2. Use the graph provided in Example 14.1 to...Ch. 14.2 - 1. Which of the following will not usually...Ch. 14.3 - The initial rate ( [NO]/ t] of the reaction of...Ch. 14.3 - The rate constant, k, at 25 C is 0.27/h for the...Ch. 14.3 - The reaction NO(g) + 1/2 Cl2(g) NOCl(g) is...Ch. 14.4 - Sucrose, a sugar, decomposes in acid solution to...Ch. 14.4 - Gaseous azomethane (CH3N2CH3) decomposes to ethane...
Ch. 14.4 - Prob. 3CYUCh. 14.4 - The catalyzed decomposition of hydrogen peroxide...Ch. 14.4 - Americium is used in smoke detectors and in...Ch. 14.4 - The decomposition of N2O5 is a first-order...Ch. 14.4 - Which of the following will confirm that the...Ch. 14.4 - 3. The equation for the decomposition of NO2(g) at...Ch. 14.5 - Prob. 1CYUCh. 14.5 - The colorless gas N2O4, decomposes to the brown...Ch. 14.5 - Prob. 1RCCh. 14.5 - Prob. 2RCCh. 14.6 - Nitrogen monoxide is reduced by hydrogen to give...Ch. 14.6 - Prob. 2CYUCh. 14.6 - One possible mechanism for the decomposition of...Ch. 14.6 - The rate equation for a reaction A + B C was...Ch. 14.6 - A reaction is believed to occur by the following...Ch. 14.6 - Prob. 1QCh. 14.6 - Prob. 2QCh. 14.6 - Prob. 3QCh. 14.6 - Prob. 4QCh. 14.6 - Prob. 5QCh. 14.6 - Determine the activation energy for the reaction...Ch. 14 - Give the relative rates of disappearance of...Ch. 14 - Give the relative rates of disappearance of...Ch. 14 - In the reaction 2 O3(g) 3 O2(g), the rate of...Ch. 14 - In the synthesis of ammonia, if [H2]/t = 4.5 104...Ch. 14 - Experimental data are listed here for the reaction...Ch. 14 - 6. Phenyl acetate, an ester, reacts with water...Ch. 14 - Using the rate equation Rate = k[A]2[B], define...Ch. 14 - A reaction has the experimental rate equation Rate...Ch. 14 - The reaction between ozone and nitrogen dioxide at...Ch. 14 - Nitrosyl bromide, NOBr, is formed from NO and Br2:...Ch. 14 - The data in the table are for the reaction of NO...Ch. 14 - The reaction 2 NO(g) + 2 H2(g) N2(g) + 2 H2O(g)...Ch. 14 - Data for the reaction NO(g) + O2(g) NO2(g) are...Ch. 14 - Data for the following reaction are given in the...Ch. 14 - The rate equation for the hydrolysis of sucrose to...Ch. 14 - The decomposition of N2O5 in CCl4 is a first-order...Ch. 14 - The decomposition of SO2Cl2 is a first-order...Ch. 14 - The conversion of cyclopropane to propene (Example...Ch. 14 - Hydrogen peroxide, H2O2(aq), decomposes to H2O()...Ch. 14 - The decomposition of nitrogen dioxide at a high...Ch. 14 - At 573 K, gaseous NO2(g) decomposes, forming NO(g)...Ch. 14 - The dimerization of butadiene, C4H6, to form...Ch. 14 - The decomposition of ammonia on a metal surface to...Ch. 14 - Hydrogen iodide decomposes when heated, forming...Ch. 14 - The rate equation for the decomposition of N2O5...Ch. 14 - Gaseous azomethane, CH3N=NCH3, decomposes in a...Ch. 14 - The decomposition of SO2Cl2 SO2Cl2(g) SO2(g) +...Ch. 14 - The compound Xe(CF3)2 decomposes in a first-order...Ch. 14 - The radioactive isotope 64Cu is used in the form...Ch. 14 - Radioactive gold-198 is used in the diagnosis of...Ch. 14 - Prob. 31PSCh. 14 - Ammonia decomposes when heated according to the...Ch. 14 - Gaseous NO2 decomposes at 573 K. NO2(g) NO(g) + ...Ch. 14 - The decomposition of HOF occurs at 25 C. HOF(g) ...Ch. 14 - Prob. 35PSCh. 14 - Prob. 36PSCh. 14 - Calculate the activation energy, Ea, for the...Ch. 14 - If the rate constant for a reaction triples when...Ch. 14 - When healed lo a high temperature, cyclobutane,...Ch. 14 - When heated, cyclopropane is converted to propene...Ch. 14 - The reaction of H2 molecules with F atoms H2(g) +...Ch. 14 - Prob. 42PSCh. 14 - What is the rate law for each of the following...Ch. 14 - What is the rate law for each of the following...Ch. 14 - Ozone, O3, in the Earths upper atmosphere...Ch. 14 - The reaction of NO2(g) and CO(g) is thought to...Ch. 14 - A proposed mechanism for the reaction of NO2 and...Ch. 14 - The mechanism for the reaction of CH3OH and HBr is...Ch. 14 - A reaction has the following experimental rate...Ch. 14 - For a first-order reaction, what fraction of...Ch. 14 - Prob. 51GQCh. 14 - Data for the following reaction are given in the...Ch. 14 - Formic acid decomposes at 550 C according to the...Ch. 14 - Isomerization of CH3NC occurs slowly when CH3NC is...Ch. 14 - When heated, tetrafluoroethylene dimerizes to form...Ch. 14 - Data in the table were collected at 540 K for the...Ch. 14 - Ammonium cyanate, NH4NCO, rearranges in water to...Ch. 14 - Prob. 58GQCh. 14 - At temperatures below 500 K, the reaction between...Ch. 14 - Nitryl fluoride can be made by treating nitrogen...Ch. 14 - The decomposition of dinitrogen pentaoxide N2O5(g)...Ch. 14 - The data in the table give the temperature...Ch. 14 - The decomposition of gaseous dimethyl ether at...Ch. 14 - The decomposition of phosphine, PH3, proceeds...Ch. 14 - The thermal decomposition of diacetylene, C4H2,...Ch. 14 - Prob. 66GQCh. 14 - The ozone in the Earths ozone layer decomposes...Ch. 14 - Hundreds of different reactions occur in the...Ch. 14 - Data for the reaction [Mn(CO)5(CH3CN)]+ + NC5H5 ...Ch. 14 - The gas-phase reaction 2 N2O5(g) 4 NO2(g) + O2(g)...Ch. 14 - Prob. 71GQCh. 14 - The decomposition of SO2Cl2 to SO2 and Cl2 is...Ch. 14 - The decomposition of nitrogen dioxide at a high...Ch. 14 - Prob. 74GQCh. 14 - Egg protein albumin is precipitated when an egg is...Ch. 14 - A The compound 1,3-butadiene (C4H6) forms...Ch. 14 - Hypofluorous acid, HOF, is very unstable,...Ch. 14 - We know that the decomposition of SO2Cl2 is...Ch. 14 - Nitramide, NO2NH2, decomposes slowly in aqueous...Ch. 14 - Prob. 80GQCh. 14 - Prob. 83ILCh. 14 - Prob. 84ILCh. 14 - The oxidation of iodide ion by the hypochlorite...Ch. 14 - The acid-catalyzed iodination of acetone...Ch. 14 - Prob. 87SCQCh. 14 - The following statements relate to the reaction...Ch. 14 - Chlorine atoms contribute to the destruction of...Ch. 14 - Prob. 91SCQCh. 14 - Prob. 92SCQCh. 14 - The reaction cyclopropane propene occurs on a...Ch. 14 - Prob. 94SCQCh. 14 - Examine the reaction coordinate diagram given...Ch. 14 - Draw a reaction coordinate diagram for an...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the reaction 4NO2(g)+O2(g)2N2O5(g) (a) express the rate of formation of N2O5 in terms of the rate of disappearance of O2. (b) suppose the rate of disappearance of O2 is 0.0037 mol L1 s1. Calculate the rate of disappearance of NO2.arrow_forward11.93 On a particular day, the ozone level in Milwaukee exceeded the EPAs 1-hour standard of 0.12 ppin by 10 ppb. How many ozone molecules would be present in 1 liter of air at the detection site?arrow_forwardWhen boron trifluoride reacts with ammonia, the following reaction occurs: BF3(g)+NH3(g)BF3NH3(g)The following data are obtained at a particular temperature: (a) What is the order of the reaction with respect to BF3, NH3, and overall? (b) Write the rate expression for the reaction. (c) Calculate k for the reaction. (d) When [ BF3 ]=0.533M and NH3=0.300M, what is the rate of the reaction at the temperature of the experiment?arrow_forward
- Hydrogen bromide is a highly reactive and corrosive gas used mainly as a catalyst for organic reactions. It is produced by reacting hydrogen and bromine gases together. Â H2(g)+Br2(g)2HBr(g)The rate is followed by measuring the intensity of the orange color of the bromine gas. The following data are obtained: (a) What is the order of the reaction with respect to hydrogen, bromine, and overall? (b) Write the rate expression of the reaction. (c) Calculate k for the reaction. What are the units for k? (d) When [ H2 ]=0.455Mand [ Br2 ]=0.215M, what is the rate of the reaction?arrow_forward11.41 For a drug to be effective in treating an illness, its levels in the bloodstream must be maintained for a period of time. One way to measure the level of a drug in the body is to measure its rate of appearance in the urine. The rate of excretion of penicillin is first order, with a half-life of about 30 min. If a person receives an injection of 25 mg of penicillin at t = 0, how much penicillin remains in the body after 3 hours?arrow_forwardThe decomposition of sulfuryl chloride, SO2Cl2, to sulfur dioxide and chlorine gases is a first-order reaction. SO2Cl2(g)SO2(g)+Cl2(g)At a certain temperature, the half-life of SO2Cl2 is 7.5102 min. Consider a sealed flask with 122.0 g of SO2Cl2. (a) How long will it take to reduce the amount of SO2Cl2 in the sealed flask to 45.0 g? (b) If the decomposition is stopped after 29.0 h, what volume of Cl2 at 27C and 1.00 atm is produced?arrow_forward
- Express the rate of the reaction 2N2O(g)2N2(g)+O2(g) in terms of (b) [ N2O ] (a) [ O2 ]arrow_forwardThe rate of the decomposition of hydrogen peroxide, H2O2, depends on the concentration of iodide ion present. The rate of decomposition was measured at constant temperature and pressure for various concentrations of H2O2and of KI. The data appear below. Determine the order of reaction for each substance, write the rate law, and evaluate the rate constant. Rate [H2OJ [Kll (mL min-’) (mol L ’) (mol L ’) 0.090 0.15 0.033 0.178 0.30 0.033 0.184 0.15 0.066arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY