
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.4, Problem 14.11P
What products would you expect from oxidation of the following alcohols?
(a) CH3CH2CH2OH
(b) 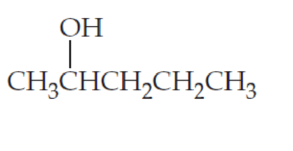
(c) 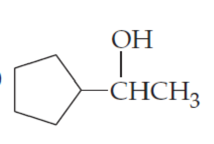
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What disulfides would you obtain from oxidation of the following thiols?(a) CH3CH2CH2SH (b) 3-Methylbutane-1-thiol (skunk scent)
When pentane is exposed to Br2 in the presence of light, a halogenation reaction occurs. Write the formulas of:(a) All possible products containing only one bromine(b) All possible products containing two bromines that are not on the same carbon
Propose a reasonable mechanism, using curved arrow notation, for the reaction performed in this experiment. Your answer should include the following:(1) Decomposition of the 3-sulfolene(2) cycloaddition of 1,3-butadiene with maleic anhydride including the HOMO-LUMO interactions(3) hydrolysis of the anhydride under BASE hydrolysis with NaOH in H2O (consider how this differs from what was done in the experiment)Check attached image.
Chapter 14 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 14.1 - Identify each of the following compounds as an...Ch. 14.1 - Prob. 14.2PCh. 14.2 - Prob. 14.3PCh. 14.2 - Prob. 14.4PCh. 14.2 - Prob. 14.5PCh. 14.3 - Prob. 14.6PCh. 14.3 - For each of the following molecules, (i) redraw...Ch. 14.4 - Prob. 14.1MRPCh. 14.4 - Provide the mechanism for the dehydration of...Ch. 14.4 - Prob. 14.3MRP
Ch. 14.4 - Prob. 14.8PCh. 14.4 - What alcohols yield the following alkenes as the...Ch. 14.4 - Prob. 14.10KCPCh. 14.4 - What products would you expect from oxidation of...Ch. 14.4 - Prob. 14.12PCh. 14.4 - Prob. 14.13KCPCh. 14.5 - Prob. 14.14PCh. 14.5 - Prob. 14.15PCh. 14.7 - Prob. 14.1CIAPCh. 14.7 - Prob. 14.2CIAPCh. 14.7 - Prob. 14.3CIAPCh. 14.7 - Prob. 14.16PCh. 14.8 - What disulfides would you obtain from oxidation of...Ch. 14.9 - Prob. 14.18PCh. 14.10 - Prob. 14.19PCh. 14.10 - Prob. 14.20PCh. 14.10 - Prob. 14.4CIAPCh. 14.10 - Prob. 14.5CIAPCh. 14.10 - Prob. 14.6CIAPCh. 14.10 - Prob. 14.7CIAPCh. 14 - Prob. 14.21UKCCh. 14 - Prob. 14.22UKCCh. 14 - Prob. 14.23UKCCh. 14 - Prob. 14.24UKCCh. 14 - Prob. 14.25UKCCh. 14 - How do alcohols, ethers, and phenols differ...Ch. 14 - What is the structural difference between primary,...Ch. 14 - Prob. 14.28APCh. 14 - Prob. 14.29APCh. 14 - The Taxane nucleus is shown here; it is the basis...Ch. 14 - Vitamin E has the structure shown. Identify the...Ch. 14 - Give systematic names for the following alcohols:...Ch. 14 - Give systematic names for the following compound...Ch. 14 - Draw structures corresponding to the following...Ch. 14 - Draw structures corresponding to the following...Ch. 14 - Prob. 14.36APCh. 14 - Locate the alcohol functional groups in the taxane...Ch. 14 - Prob. 14.38APCh. 14 - Prob. 14.39APCh. 14 - Prob. 14.40APCh. 14 - Prob. 14.41APCh. 14 - Prob. 14.42APCh. 14 - Prob. 14.43APCh. 14 - Assume that you have samples of the following two...Ch. 14 - Which of the following alcohols can undergo...Ch. 14 - The following alkenes can be prepared by...Ch. 14 - Prob. 14.47APCh. 14 - Prob. 14.48APCh. 14 - What alcohols would you oxidize to obtain the...Ch. 14 - Prob. 14.50APCh. 14 - What is the structural relationship between a...Ch. 14 - Prob. 14.52APCh. 14 - Prob. 14.53APCh. 14 - Prob. 14.54APCh. 14 - Prob. 14.55APCh. 14 - Prob. 14.56APCh. 14 - Prob. 14.57APCh. 14 - Identify the chiral center(s) in each of the...Ch. 14 - Are the following molecules chiral or achiral? If...Ch. 14 - Prob. 14.60CPCh. 14 - Prob. 14.61CPCh. 14 - 1-Propanol is freely soluble in water, 1-butanol...Ch. 14 - Prob. 14.63CPCh. 14 - Prob. 14.64CPCh. 14 - Prob. 14.65CPCh. 14 - Prob. 14.66CPCh. 14 - Prob. 14.67CPCh. 14 - Prob. 14.68CPCh. 14 - Prob. 14.69CPCh. 14 - Prob. 14.70CPCh. 14 - Prob. 14.71CPCh. 14 - Prob. 14.72CPCh. 14 - (a)Draw all possible cyclic C7H14O alcohol isomers...Ch. 14 - Prob. 14.74GPCh. 14 - Prob. 14.75GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the major organic product of the following reaction: 1. (CH3CH2)2NH, H+ (cat.) 2. о H 3. H3O+arrow_forwardThe decomposition of various substituted benzyl azoxyarenesulfonates in trifluoroethanol was studied. The overall reaction is shown below F3C OH CF3 HO,S + N20 The decomposition of various substituted benzyl azoxyarenesulfonates in trifluoroethanol was studied. The overall reaction is shown below F,C OH HO,S + N,0 Y kobs (S ') 3-CI CH3 3.1 x 10-7 4-CI CH3 2.3 х 10-6 3-CH3 4-CH3 4-OCH3 CH3 CHs 4.7х 10-6 CH3 7.6х 10-6 CH3 6.0 x 10-5 1.7 x 10-3 4-CH OCH3 4.24 x 10-5 4-CH3 4-CH3 4-CH3 CH3 Br 6.0 x 10-5 1.91 x 10-4 CN 4.65 x 10-4 a. Using the data provided, prepare Hammett plots to quantify the effects of the substituents X and Y on the reaction's rate. Attach them to your problem set. b. Based on your Hammett analysis, propose a complete, curved-arrow mechanism for this reaction. Use a sentence or two to explain how your Hammett analyses support your proposed mechanism.arrow_forwardWhich of the following classifications apply to the formation of 3-hydroxybutyrate from acetoacetate?(a) Condensation (b) Hydrolysis(c) Oxidation (d) Reductionarrow_forward
- In the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forwardDraw the structure of two different aldohexoses that yield the followingaldaric acid when oxidized with HNO3. Use Figure to name eachaldohexosearrow_forwardwhich statement is true for the following reaction?arrow_forward
- Draw the alcohol used to produce each of the following ketones. Include all hydrogen atoms.arrow_forwardWhat structural feature is necessary for an alcohol to undergo oxidation reactions?arrow_forward1) a) Write the mechanism for the reaction of 2-phosphoglycerate yielding phosphoenolpyruvate.Include important resonance structures.b) In the enolase enzyme reaction, OH–is a very poor leaving group. How is this handled inthis biological system?arrow_forward
- In observing a Haworth or cyclohexane-chair representation of alpha-d-glucopyranose, the anomeric carbon can de best identified by: a) the carbon atom which is bonded to the most H atoms b) the carbon atom which is bonded to the least H atoms c) The carbon atom which has its OH group pointing down d) The carbon atom which is bonded to two oxygen atoms Cellulose differs from amylose in that: a) cellulose has 1-6 branches while amylose does not b) amylose has 1-6 branches while cellulose does not c) amylose has alpha glycosidic bonds while cellulose has beta glycosidic bonds d) cellulose contains sulfate while amylose does not. The glycosaminoglycan (mucopolysaccharide) which is not normally associated with joint tissue is: a) heparin b) keratan sulfate c) chondroitin sulfate d) hyaluronic acid In glycoproteins, which amino acid does not bond sugar molecules? a) Asn b) Ser c) Thr d) Gly Which feature do all lipid molecules share in common? a) fatty acid molecules…arrow_forwardWhich of these statements concerning this reaction is true? acetaldehyde + NADH ↔ ethanol + NAD+ a) Ethanol is more reduced than acetaldehyde. b) Acetaldehyde is more reduced than ethanol. c) NAD+ is more reduced than NADH. d) Ethanol is more oxidized than acetaldehyde.arrow_forwardIn the electrolysis of aqueous sodium bromide, there are two possible anodic reactions: *2H2O(l) ——-> 02(g) + 4H+(aq) + 4e–, E° = 1.23V2Br–(aq) ——–> Br2(g) + 2e-2, E° = 1.08 VWhich reaction occurs at anode and why?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Biodiversity hotspots and functional diversity; Author: Stockholm Resilience Centre TV;https://www.youtube.com/watch?v=Gr_eIsFOKr4;License: Standard Youtube License