
Concept explainers
(a)
Interpretation:
The carbonyl containing product should be identified when the given alcohol undergoes oxidation reaction.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
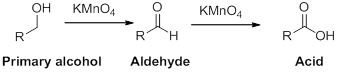
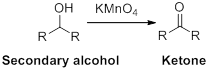
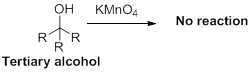
(b)
Interpretation:
The carbonyl containing product should be identified when the given alcohol undergoes oxidation reaction.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
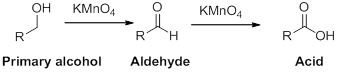
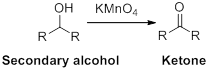
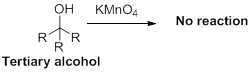
(c)
Interpretation:
The product should be identified when it is undergoes oxidation reaction.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
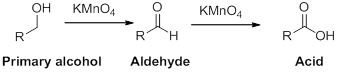
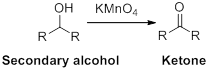
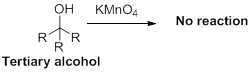
(d)
Interpretation:
The carbonyl containing product should be identified when the given alcohol undergoes oxidation reaction.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
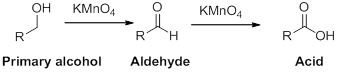
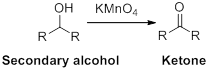
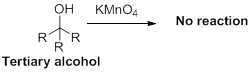
(e)
Interpretation:
The carbonyl containing product should be identified when the given alcohol undergoes oxidation reaction.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
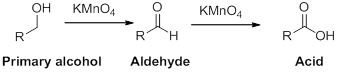
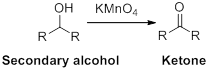
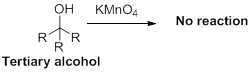
(b)
Interpretation:
The carbonyl containing product should be identified when the given alcohol undergoes oxidation reaction.
Concept introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
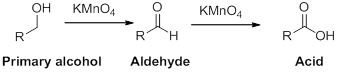
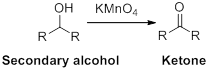
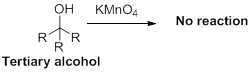
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Propose a reasonable mechanism, using curved arrow notation, for the reaction performed in this experiment. Your answer should include the following:(1) Decomposition of the 3-sulfolene(2) cycloaddition of 1,3-butadiene with maleic anhydride including the HOMO-LUMO interactions(3) hydrolysis of the anhydride under BASE hydrolysis with NaOH in H2O (consider how this differs from what was done in the experiment)Check attached image.arrow_forwardDraw a Fischer projection formula for the enantiomer of each of the following monosaccharides. (a to d)arrow_forwardWhat are the expected results for the reactions: (a) CuSO4 with iron, (b) KMnO4 with ethanol, and (c) KMnO4 with acetaldehyde? Write the chemical equation of the reactions, and identify which substance is reduced and which is oxidized.arrow_forward
- When pentane is exposed to Br2 in the presence of light, a halogenation reaction occurs. Write the formulas of:(a) All possible products containing only one bromine(b) All possible products containing two bromines that are not on the same carbonarrow_forwardPotentiometric titration curve is given below, which is obtained during the potentiometric titration between strong base KOH (0.2 M) with strong acid HI, label the point in the curve from the following options. If more than one points are present than write as x, y(means separate by using comma) a)The point where pH is because of excess OH - ions. b) The point where pH is only because of HI in water. c)The point where [HI]= [I] in water. d)The point where pH=pka e) The point where all HI is neutralized. f) The point where pH corresponds to solution of [I- ] in water. 14 13 12 11 10 9 pH 6. TITIT TITarrow_forwardDraw the structure of two different aldohexoses that yield the followingaldaric acid when oxidized with HNO3. Use Figure to name eachaldohexosearrow_forward
- What disulfides would you obtain from oxidation of the following thiols?(a) CH3CH2CH2SH (b) 3-Methylbutane-1-thiol (skunk scent)arrow_forwardIf Taxol (see Problem ) has a specific rotation of -49°, then what is the specific rotation of its enantiomer?arrow_forwardDrawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forward
- Fumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forwardwhich statement is true for the following reaction?arrow_forward(a) Label all the O atoms that are part of a glycoside in rebaudioside A. Rebaudioside A, marketed under the trade name Truvia, is a sweet glycoside obtained from the stevia plant, which has been used for centuries in Paraguay to sweeten foods. (b) The alcohol or phenol formed from the hydrolysis of a glycoside is called an aglycon. What aglycon and monosaccharides are formed by the hydrolysis of rebaudioside A?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





