
Concept explainers
Interpretation:
The reason should be explained for glucose is more soluble than 1-hexanol.
Concept introduction:
Solubility of the alcohol in water:
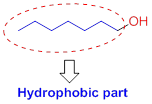
The solubility of the alcohol is mainly depends on the hydrophilic part as well as length of the alkyl chain. If the compound has more number of hydroxyl group or less number of carbon chain which is soluble in water. If the compound has less number of hydroxyl group with more number of carbon chain which is insoluble in water because of hydrophobic part.
Alcohols have two different parts
- (1) Hydrophilic (water-loving),
- (2) Hydrophobic (water-fearing).
Hydrophilic (water-loving):
The compound has the hydroxyl group (

Hydrophobic (water-fearing):
The compound has alkyl chain group is called as hydrophilic.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Using the tree structures for the following monosaccharides and comparing to that for glucose, draw the structures of the following: A. tree (Fischer) structures of: D-mannose and L-mannose B. ring structures of alpha-D-mannose, beta-D-mannose, alpha-L-mannose, and beta-L-mannose Draw the ring (Haworth) structures as 6-membered pyranoses, being careful to show the positions of the hydroxyl groups above or below the plane of the ring.arrow_forwardFor a sugar to behave as a reducing sugar, it must have a free aldehyde group. Fructose is a ketose, yet it behaves like a reducing sugar. Explain.arrow_forwardMild hydrolysis of a naturally occurring parent lipid with dilute NaOH generated L-glycerol 3-phosphoserine and the sodium salt of hexadecanoate and Δ9-octadecenoate. Draw the structure of the parent lipid.arrow_forward
- write the structure of the following disaccharides ex: Beta- D- Galactopyranosyl- alpha-D-glucosearrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forwardD- and L- designations are used to distinguish between the two possible enantiomers of the monosaccharide, galactose. From the Fischer projection, determine the designation of this monosaccharide.arrow_forward
- Spermaceti, a fragrant substance isolated from sperm whales, was commonly used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester of palmiticacid with cetyl alcohol (the straight-chain 16-carbon alcohol). Draw the structure of spermaceti.arrow_forwardDraw condensed structural formulas for all products obtained from the complete hydrolysis of the following triacylglycerol.arrow_forwardCellulose is virtually insoluble in water, whereas amylose isrelatively soluble. Compare their structures and explain thisdisparity in solubility.arrow_forward
- Draw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forwardAn unknown trisaccharide was treated with dimethyl sulfate which adds a methyl group to every free OH group. It was then subjected to acid hydrolysis (to break glycoside bonds). The products were 23, 4, 6-tetra-O-methylgalactosc, 23,4-ui-O methyl glucose, and 2,3.6-tri-O methylglucose. Treatment of the intact trisaccharide with 0(1 6)-gaiactosidase yielded galactose and a saccharide. Treatment of this disaccharide with a(l 4)-glucosidase yielded D glucose. Draw the structure of this trisaccharide and give its systematic name.arrow_forwardIs D-2-deoxygalactose the same chemical as D-2-deoxyglucose? Explain.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





