
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 14.5, Problem 14.14P
(a)
Interpretation Introduction
Interpretation:
The structure should be drawn for 2,4-Dinitrophenol.
Concept Introduction:
- (1) Substituted benzenes are named using ‘-benzene’ as the parent.
- (2) When benzene has more than one substituent, the position of those substituents is indicated by numbers or by ortho(o-), meta(m-) or para(p-).
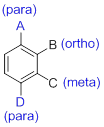
(b)
Interpretation Introduction
Interpretation:
The structure should be drawn for m-ethyl phenol.
Concept Introduction:
- (1) Substituted benzenes are named using ‘-benzene’ as the parent.
- (2) When benzene has more than one substituent, the position of those substituents is indicated by numbers or by ortho(o-), meta(m-) or para(p-).

Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-ol
Draw structures of the following compounds:(a) cis-Hept-3-ene(b) cis-4-Methylpent-2-ene(c) trans-2,5-Dimethylhex-3-ene
Draw structures corresponding to the following names:(a) 3-Methylhexan-1-ol (b) 1-Methyl-3-propylcyclopentanol(c) 2,2-Dimethylhexan-3-ol (d) Heptan-3-ol(e) 2,3-Diethylcyclohexanol
Chapter 14 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 14.1 - Identify each of the following compounds as an...Ch. 14.1 - Prob. 14.2PCh. 14.2 - Prob. 14.3PCh. 14.2 - Prob. 14.4PCh. 14.2 - Prob. 14.5PCh. 14.3 - Prob. 14.6PCh. 14.3 - For each of the following molecules, (i) redraw...Ch. 14.4 - Prob. 14.1MRPCh. 14.4 - Provide the mechanism for the dehydration of...Ch. 14.4 - Prob. 14.3MRP
Ch. 14.4 - Prob. 14.8PCh. 14.4 - What alcohols yield the following alkenes as the...Ch. 14.4 - Prob. 14.10KCPCh. 14.4 - What products would you expect from oxidation of...Ch. 14.4 - Prob. 14.12PCh. 14.4 - Prob. 14.13KCPCh. 14.5 - Prob. 14.14PCh. 14.5 - Prob. 14.15PCh. 14.7 - Prob. 14.1CIAPCh. 14.7 - Prob. 14.2CIAPCh. 14.7 - Prob. 14.3CIAPCh. 14.7 - Prob. 14.16PCh. 14.8 - What disulfides would you obtain from oxidation of...Ch. 14.9 - Prob. 14.18PCh. 14.10 - Prob. 14.19PCh. 14.10 - Prob. 14.20PCh. 14.10 - Prob. 14.4CIAPCh. 14.10 - Prob. 14.5CIAPCh. 14.10 - Prob. 14.6CIAPCh. 14.10 - Prob. 14.7CIAPCh. 14 - Prob. 14.21UKCCh. 14 - Prob. 14.22UKCCh. 14 - Prob. 14.23UKCCh. 14 - Prob. 14.24UKCCh. 14 - Prob. 14.25UKCCh. 14 - How do alcohols, ethers, and phenols differ...Ch. 14 - What is the structural difference between primary,...Ch. 14 - Prob. 14.28APCh. 14 - Prob. 14.29APCh. 14 - The Taxane nucleus is shown here; it is the basis...Ch. 14 - Vitamin E has the structure shown. Identify the...Ch. 14 - Give systematic names for the following alcohols:...Ch. 14 - Give systematic names for the following compound...Ch. 14 - Draw structures corresponding to the following...Ch. 14 - Draw structures corresponding to the following...Ch. 14 - Prob. 14.36APCh. 14 - Locate the alcohol functional groups in the taxane...Ch. 14 - Prob. 14.38APCh. 14 - Prob. 14.39APCh. 14 - Prob. 14.40APCh. 14 - Prob. 14.41APCh. 14 - Prob. 14.42APCh. 14 - Prob. 14.43APCh. 14 - Assume that you have samples of the following two...Ch. 14 - Which of the following alcohols can undergo...Ch. 14 - The following alkenes can be prepared by...Ch. 14 - Prob. 14.47APCh. 14 - Prob. 14.48APCh. 14 - What alcohols would you oxidize to obtain the...Ch. 14 - Prob. 14.50APCh. 14 - What is the structural relationship between a...Ch. 14 - Prob. 14.52APCh. 14 - Prob. 14.53APCh. 14 - Prob. 14.54APCh. 14 - Prob. 14.55APCh. 14 - Prob. 14.56APCh. 14 - Prob. 14.57APCh. 14 - Identify the chiral center(s) in each of the...Ch. 14 - Are the following molecules chiral or achiral? If...Ch. 14 - Prob. 14.60CPCh. 14 - Prob. 14.61CPCh. 14 - 1-Propanol is freely soluble in water, 1-butanol...Ch. 14 - Prob. 14.63CPCh. 14 - Prob. 14.64CPCh. 14 - Prob. 14.65CPCh. 14 - Prob. 14.66CPCh. 14 - Prob. 14.67CPCh. 14 - Prob. 14.68CPCh. 14 - Prob. 14.69CPCh. 14 - Prob. 14.70CPCh. 14 - Prob. 14.71CPCh. 14 - Prob. 14.72CPCh. 14 - (a)Draw all possible cyclic C7H14O alcohol isomers...Ch. 14 - Prob. 14.74GPCh. 14 - Prob. 14.75GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Give structures for the following, based on the data in Table . (a) cis-9-Dodecenoic acid (b) 18:1c∆11 (c) A saturated fatty acid that should melt below 30 °Carrow_forwardDraw structures corresponding to the following names:(a) 2,4-Dimethylheptan-2-ol(b) 2,2-Diethylcyclohexanol(c) 5-Ethyl-5-methylheptan-1-ol(d) 4-Ethylhexan-2-ol(e) 3-Methoxycyclooctanol(f) 3,3-Dimethylheptane-1,6-diolarrow_forwardSpermaceti, a fragrant substance isolated from sperm whales, was commonly used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester of palmiticacid with cetyl alcohol (the straight-chain 16-carbon alcohol). Draw the structure of spermaceti.arrow_forward
- Draw a Lewis structure for hydroxylamine, NH2OH.arrow_forwardDraw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardIndicate the following for each disaccharide: I) type of glycosidic linkage i.e. a(1→4) II) configuration i.e. alpha or beta 1. ОН CH₂OH OH I) II) III) IV) 0 ОН 0 CH₂OH OH ОН OH 2. HO CH₂OH I) II) III) IV) ОН HOCH₂, OH ·0 НО 0 ОНА CH₂OH 3. HO I) II) III) IV) CH OH OH O ОН III) reducing or non-reducing sugar IV) name of each monosaccharide HO CH₂ ОН OH ОН 4. HO I) II) III) IV) CH₂OH 0 OH ОН CH₂OH OH О ОН ОНarrow_forward
- Give structures for the following, based on the data as shown.(a) cis-9-Dodecenoic acid(b) 18 : 1cΔ11(c) A saturated fatty acid that should melt below 30 °Carrow_forwardA) Describe the glycosidic bond (using standard convention) indicated by “Arrow a.” B) Draw the open chain Fischer projection formula of the monosaccharide labeled “B” C) Describe the glycosidic bond (as in question A) indicated by “Arrow b.”arrow_forwardIndicate the following for each disaccharide: I) type of glycosidic linkage i.e. a(1→4) II) configuration i.e. alpha or beta 1. OH CH₂OH 0 OH I) II) III) IV) OH CH₂OH -0 OH ОН OH 2. HO CH₂OH 0 I) II) III) IV) ОН HOCH2 ОН HO ОН CH₂OH 3. CH, OH CH2 О CG OH ОН HO ОН HỎ I) II) III) III) reducing or non-reducing sugar IV) name of each monosaccharide IV) OH OH 4. HO I) II) III) IV) CH₂OH OH 0 ОН CH₂OH OH ОН OHarrow_forward
- Identify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forwardDraw the keto or enol tautomer for each of the following compounds when they were treated with traces of acids or bases. CH3 OH (1) ? H H (2) CH₂CH₂ C=C₂ CH3 ?arrow_forwardDraw the two possible Haworth structures (both alpha and beta anomers) for the following monosaccharides and give their corresponding systematic names. [Show the stepwise process]arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license