
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.4, Problem 14.7P
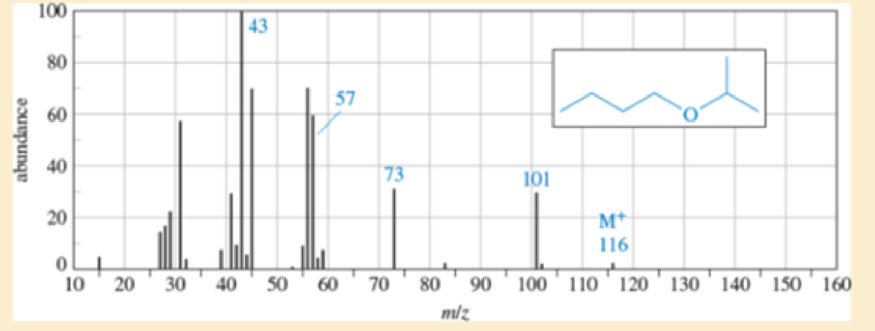
Propose a fragmentation to account for each numbered peak in the mass spectrum of n-butyl isopropyl ether.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the fragments in this mass spectrum of 2,2-dimethylbutane
How would you differentiate between 1-methoxybutane and 2-methoxy-2-methylpropane using mass spectrometry. Draw a pridictive mass spectra for bothcompounds showing the major fragmentations including possible homocleavages,heterocleavages, base peak and molecular ion.
EXPLAIN differences in fragmentation in isomers
The following two mass spectra represent 1-bromo-4-ethylbenzene and (1-bromoethyl)benzene, respectively.
Chapter 14 Solutions
Organic Chemistry (9th Edition)
Ch. 14.2C - Rank the given solvents in decreasing order of...Ch. 14.2D - Prob. 14.2PCh. 14.2D - In the presence of 18-crown-6, potassium...Ch. 14.3B - Give a common name (when possible) and a...Ch. 14.3C - Prob. 14.5PCh. 14.3C - Name the following heterocyclic ethers.Ch. 14.4 - Propose a fragmentation to account for each...Ch. 14.5 - Prob. 14.8PCh. 14.5 - Prob. 14.9PCh. 14.6 - Prob. 14.10P
Ch. 14.7 - Explain why bimolecular condensation is a poor...Ch. 14.7 - Prob. 14.12PCh. 14.7 - Prob. 14.13PCh. 14.8 - Prob. 14.14PCh. 14.8 - Prob. 14.15PCh. 14.8 - Prob. 14.16PCh. 14.10A - Prob. 14.17PCh. 14.10A - Prob. 14.18PCh. 14.10B - Prob. 14.19PCh. 14.11B - Show how you would accomplish the following...Ch. 14.11B - Prob. 14.21PCh. 14.12 - Prob. 14.22PCh. 14.12 - Prob. 14.23PCh. 14.12 - Prob. 14.24PCh. 14.13 - Prob. 14.25PCh. 14.13 - Prob. 14.26PCh. 14.14 - Prob. 14.27PCh. 14.15 - Give the expected products of the following...Ch. 14 - Write structural formulas for the following...Ch. 14 - Give common names for the following compounds. a....Ch. 14 - Give IUPAC names for the following compounds. a....Ch. 14 - Glycerol (propane-1,2,3-triol) is a viscous syrup...Ch. 14 - Prob. 14.33SPCh. 14 - Show how you would make the following ethers,...Ch. 14 - (A true story.) An inexperienced graduate student...Ch. 14 - Prob. 14.36SPCh. 14 - a. Show how you would synthesize the pure (R)...Ch. 14 - a. Predict the values of m/z and the structures of...Ch. 14 - The following reaction resembles the...Ch. 14 - Prob. 14.40SPCh. 14 - Prob. 14.41SPCh. 14 - Prob. 14.42SPCh. 14 - Give the structures of the intermediates...Ch. 14 - Prob. 14.44SPCh. 14 - Show how you would synthesize the following ethers...Ch. 14 - Prob. 14.46SPCh. 14 - Prob. 14.47SPCh. 14 - Prob. 14.48SPCh. 14 - An acid-catalyzed reaction was carried out using...Ch. 14 - Propylene oxide is a chiral molecule. Hydrolysis...Ch. 14 - Prob. 14.51SPCh. 14 - Prob. 14.52SPCh. 14 - Prob. 14.53SPCh. 14 - Prob. 14.54SPCh. 14 - In 2012, a group led by Professor Masayuki Satake...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For each structure below, use numbers to indicate chemically equivalent and distinct hydrogens,and make a table showing the predicted integration and multiplicity of each peak cluster.arrow_forwardConsider isomeric alcohols A and B and mass spectra [1] and [2].(a) Label the molecular ion and base peak in each spectrum. (b) Use the fragmentation patterns to determine which mass spectrum corresponds to isomer A and which corresponds to isomer B.arrow_forwardExplain per step. The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropaneare shown. Match the compounds with the spectra.arrow_forward
- What mass spectral fragments are formed from α cleavage of pentan-2-one,CH3COCH2CH2CH3?arrow_forwardPredict the masses and the structures of the most abundant fragments observed in the mass spectra of the followingcompounds. 3-bromo-2-methylpentanearrow_forwardEI-MS of ortho nitrotoluene shows a large fragment ion at m/z 120 .The EI-MS of Tri deuteron ortho nitrotoluene does not show any peak at m/z 120 but does have a peak at m/z 122 .Show the fragmentation pattern that accounts for the above observation.arrow_forward
- Explain per step. Given the MS spectra of 2-pentanol, show the fragmentation pattern responsible for m/z values.arrow_forwardHow could IR spectroscopy distinguish between 1,5-hexadiene and 2,4-hexadiene?arrow_forwardWhich amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]-arrow_forward
- Which amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]- i and iii only ii ii and iv only i only iiarrow_forwardPlease help me correct the peak integrations and multiplicities of the H-NMR for 1-Bromo-2-Hexene.arrow_forwardThree mass spectra were shown below, Spectrums B-D, which corresponds to either methyI benzoate, 4-methoxybenzaldehyde, or p-toluic acid. Explain which spectrum goes with which compound.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY