
(a)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
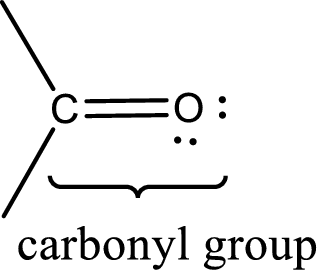
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(b)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
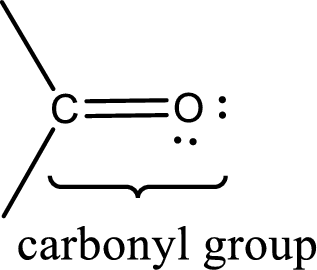
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(c)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
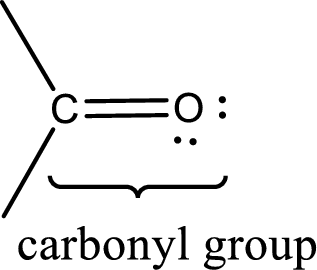
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(d)
Interpretation:
Condensed structural formula for
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
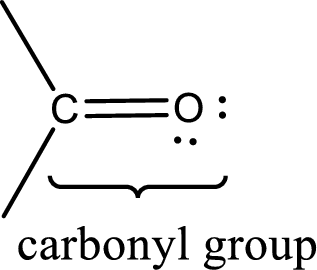
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
Trending nowThis is a popular solution!

Chapter 15 Solutions
General, Organic, and Biological Chemistry
- Draw the condensed structural formula for the fatty acid whose numerical shorthand designation is 18:2 (△ 9,12)arrow_forwardIf a monosaccharide undergoes a cyclization reaction, how do you determine the alpha and beta designation of the new product? A.check if there is a new asymmetric carbon B. L and D isoform is used as the linear reactant C. whether the C5 hydroxyl group attacks the front or back of the anomeric carbon D. whether the majority of the hydroxyl groups are above or below the plane of the ringarrow_forwardDraw an aspartic acid and label: a. The acarbon, aamino, and acarboxyl groups (you can use the chart at the end of the exam for the R group). b. At pH=11, what would be the overall charge on aspartic acid in solution? You can use the short-hand rule...arrow_forward
- A monosaccharide designated as an aldehyde sugar contains(a) a terminal carboxyl group (b) an internal carboxyl group(c) a terminal carbonyl group (d) an internal carbonyl group(e) a terminal carboxyl group and an internal carbonyl grouparrow_forwardAmylopectin has which of the following structural features? a. only α(1,4) glycosidic linkages b. only β(1,4) glycosidic linkages c. both α(1,4) and α(1,6) glycosidic linkages d. both α(1,4) and β(1,6) glycosidic linkagesarrow_forwardIndicate whether each of the following statements about the structural characteristics of monosaccharides is true or false. a. Both an aldehyde group and a ketone group are always present. b. An aldehyde group and at least two hydroxyl groups must be present.arrow_forward
- The molecular formula for glucose is C6H12O6. What wouldbe the molecular formula for a polymer made by linking tenglucose molecules together by dehydration reactions?(A) C60H120O60(B) C60H102O51(C) C60H100O50(D) C60H111O51arrow_forwardIDENTIFY THE FUNCTIONAL GROUP PRESENT IN THESE COMPOUNDS. A = ? B = ? C = ?arrow_forwardNumerous drugs are obtained from Glycosides. If you were to conduct a research using glycoside as active constituent, what type of glycoside are you going to isolate and what will be the possible indication of your investigational drug? Explain your answer in not less than 150 words.arrow_forward
- Indicate whether each of the following statements about the structural characteristics of monosaccharides is true or false. a. An aldehyde group or a ketone group, but not both, is always present. b. A hydroxyl group must be present on each carbon atom.arrow_forwardBased on the structure below: 1. Based on the chemistry, is the drug phototoxic? (Yes or No) 2. Is the drug is acid-stable? (Yes or No) 3. Which of the following is TRUE about the structure shown? A. The drug is doxycycline. B. The drug is resistant against dehydration. C. The drug forms into a lactone when dehydrated. The drug is bactericidal. D. The drug is phototoxic.arrow_forwardGiven below is the structure of tallose.Answer the following questiona. what is the maximum number of stereoisomers can tallose have?b. how many chiral carbons does it have?c. what is the configuration of tallose is it D or L sugar?d. what type of monosaccharide is it?arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
