
(a)
Interpretation:
The given structure has to be classified as an aldehyde, a ketone or neither.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
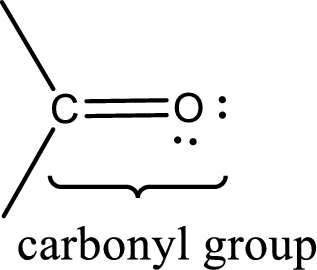
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(b)
Interpretation:
The given structure has to be classified as an aldehyde, a ketone or neither.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
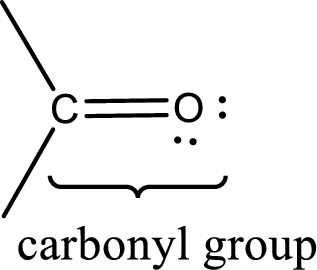
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(c)
Interpretation:
The given structure has to be classified as an aldehyde, a ketone or neither.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
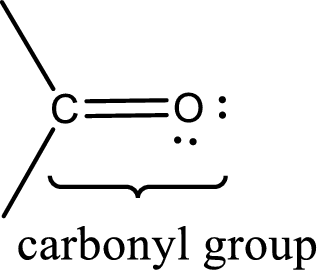
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
(d)
Interpretation:
The given structure has to be classified as an aldehyde, a ketone or neither.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
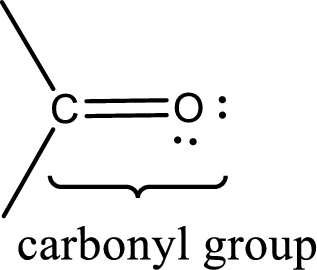
An aldehyde is a carbonyl compound in which the carbonyl carbon atom is bonded to at least one hydrogen atom directly. The other group attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
A ketone is a carbonyl compound in which the carbonyl carbon atom is bonded to two other carbon atoms directly. The groups attached to the carbonyl carbon atom can be alkyl, cycloalkyl, or aryl group.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry
- 4. Draw the line structure for the ester that can be formed from the reaction of the acid anhydride and the alcohol given below. :0: :0: HO CH3-CH2-C-o-C-CCH2-CH3 acid anhydride alcoholarrow_forward1. Which statement is correct? A. Aldehydes and ketones can hydrogen bond with other aldehydes and ketones. B. Aldehydes and ketones can hydrogen bond with water. C. Aldehydes and ketones cannot hydrogen bond with water. D. Aldehydes and ketones are always soluble in water.arrow_forwardClassify each molecule as an aldehyde, ketone, or neither. AND Classify each molecule as an ester, ether, or neither.arrow_forward
- How can phenol be distinguished from cyclohexanol? O A. solubility in water B. solubility in hydrochloric acid solution C. solubility in sodium bicarbonate solution D. solubility in sodium hydroxide solutionarrow_forwardDraw the molecular structure of an aldehyde that is an isomer of acetone.arrow_forward1. What are the properties of aldehydes and ketones? 2. What will happen to aldehyde and ketones once reduced? Oxidized?arrow_forward
- What structural characteristic is shared by the aldehydes and the ketones? A) They both are straight chain compounds. B) Aldehydes and ketones both contain a carbonyl carbon. C) Both of these compound classes have as the smallest compound a 5 carbon skeleton. D) Aldehydes and ketones have no shared characteristics.arrow_forward1. List the reasons why pentanol has a higher boiling point when compared to its corresponding alkane, pentane.arrow_forwardQUESTION 6 Classify the following as an Alcohol, Phenol, Thiol, Ether, Aldehyde or Ketone. CH2 SH H2C CH CH2-CH2 OA Alcohol O B. Phenol OC Thiol O D.Ether OE Aldehyde OF. Ketonearrow_forward
- 3. Which of the following is an ester? A. Ola B. NH O C. D.arrow_forwardComplete the following table to show what functional groups are present in each of the reagents and products in the reaction of ? (The first reagent has been filled in for you) Reagent / Product Functional group octanoic acidarrow_forwardClassify each molecule as an aldehyde, ketone, or neither.6arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

