
(a)
Interpretation:
The molecule that has higher boiling point among the given molecules has to be identified.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
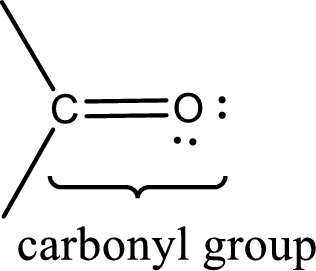
Aldehydes contain a carbonyl group that contains a hydrogen atom and a carbon atom bonded to it. Aldehyde that has one and two carbon atoms are gas at room temperature. The physical state of aldehyde that contains three carbon atoms to eleven carbon atoms that are not branched is liquid at room temperature. Aldehydes that contain more than eleven carbon atoms are solid at room temperature.
Ketones contain a carbonyl group that contains two carbon atoms bonded to it. For a compound to be ketone, a minimum of three carbon atom is required. Ketones that contain three carbon atoms to eight carbon atoms which have the carbonyl group at the second carbon atom are liquid at room temperature.
Boiling point is the temperature at which the liquid state gets converted into gaseous state. Aldehydes and ketones have their boiling points intermediate between the boiling points of alcohols and
(b)
Interpretation:
The molecule that has higher boiling point among the given molecules has to be identified.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
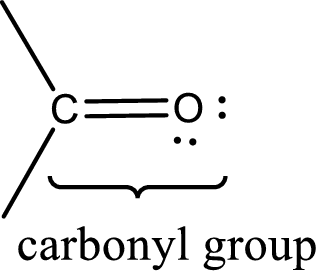
Aldehydes contain a carbonyl group that contains a hydrogen atom and a carbon atom bonded to it. Aldehyde that has one and two carbon atoms are gas at room temperature. The physical state of aldehyde that contains three carbon atoms to eleven carbon atoms that are not branched is liquid at room temperature. Aldehydes that contain more than eleven carbon atoms are solid at room temperature.
Ketones contain a carbonyl group that contains two carbon atoms bonded to it. For a compound to be ketone, a minimum of three carbon atom is required. Ketones that contain three carbon atoms to eight carbon atoms which have the carbonyl group at the second carbon atom are liquid at room temperature.
Boiling point is the temperature at which the liquid state gets converted into gaseous state. Aldehydes and ketones have their boiling points intermediate between the boiling points of alcohols and alkanes with similar molecular mass. Due to the dipole‑dipole interaction, aldehydes and ketones have higher boiling point than alkanes. Due to the absence of hydrogen bonding in aldehyde and ketones, they have lower boiling points than alcohols of similar molecular mass.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry
- i. Why is the boiling point of the aldehyde greater than that of the alkane? ii. Why is the boiling point of alcohol the highest? iii. Explain why the solubility of aldehydes and alcohols falls as the molecules get bigger.arrow_forward1. What is the most dominant intermolecular forces of attraction that exist between: a. water and limonene, the major monoterpene component of lemon essential oil? b. water and constituent ions of sodium chloride? 2. Based on your answers in number 1, which pair (water-limonene or water-sodium chloride) exhibits a stronger attraction between their molecules/ions? 3. Describe what happens during the salting-out process in terms of the intermolecular forces of attraction involved as mentioned in your previous answers.arrow_forwardwhat liquid would you expect to have the highest boiling point and why? pentane , 2-pentanol or pentanal?arrow_forward
- Industiral Chemistry Both Tetraethyl lead and methyl-t-butyl ether have been removed from or in the process of being removed from the gasoline additive industry. Explain why each of these compounds were used so highly, and why each is considered dangerous.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forward. What is the simplest aromatic alcohol commonly called? What is it mostly used for in the United States?arrow_forward
- What is the IUPAC name of the structure? a. propyl methanoate b. methyl propanoate c. propanoic acid d. 2-methyl propanoic acidarrow_forwardWhat is the IUPAC name of the structure? a. ethanoic acid b. butanoic acid c. methyl propanoate d. ethyl propanoatearrow_forwardThis carboxylic salts are effective ingredient against yeast and molds in beverages, jams, pie fillings and ketchup a. benzoates b. sorbates c. acetates d. propionatesarrow_forward
- 1. WRITE THE IUPAC NAME OF THIS ETHER CH3CH2-O-CH2CH2CH2CH2CH3 2. HOW DO PHENOLS DIFFER FROM ALCOHOLS IN TERMS OF STRUCTURE AND PROPERTIES? 3. HOW DO PHENOLS DEIFFER IN PROPERTIES FROM AROMATIC HYDROCARBONS?arrow_forwardWhat kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholsarrow_forwardThe IUPAC name of the molecule attached is? A) methylpropylmethanal B) butoxymethanal C) butoxymethanoic acid D) oxopropoxyethane E) dimethylethyl methanoate F) none of the above are correctarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


