
Interpretation:
In the given figure, the regions where multiple
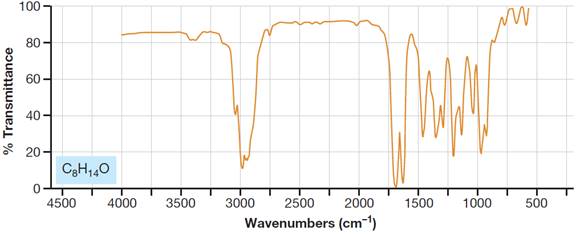
Concept introduction:
IR spectroscopy enables identification of different
One of the greatest advantages of IR spectroscopy is that the frequency of a particular type of vibration is typically found within a characteristic range of frequencies, regardless of the functional group with which the vibration is associated. The fact that absorptions by certain vibrations appear with characteristic frequency ranges enables us to use IR spectroscopy to obtain structural information about a molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Based on the spectra you located, does your molecule have a carbonyl? If so, what functional group is it a part of (carboxylic acid, ketone, aldehyde, ester, amide) and what is the frequency (in wavenumbers) of the absorption peak? If not, what is the approximate frequency range for a carbonyl? Does your molecule have either an –O-H or –N-H bond? If so, what functional group is it a part of (carboxylic acid, alcohol, amine, amide) and what is the frequency (in wavenumbers) of the absorption peak? If not, what are the approximate frequency ranges for an –O-H and an –N-H bond? Does your molecule have either an alkyne or nitrile functional group? If so, which functional group is it and what is the frequency (in wavenumbers) of the absorption peak? If not, what is the approximate frequency range for a triple bond?arrow_forwardWhat does cross peak X in Figure tell youarrow_forwardPlease help me answer letter B, number 1,2 and 3arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

