
Concept explainers
Interpretation:
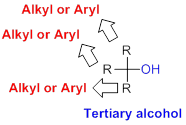
The alcohol synthesised formed
Concept Introduction:
Oxidation and Reduction: The several organic molecules oxidation is a process by which a carbon atom gains to more electronegative elements most commonly oxygen, the reduction process by which a carbon atom gains bonds to less electronegative elements.
Reduction Reaction: This process in which any substance atoms, ion or molecule gains one or more electrons it is called reduction.
Tertiary alcohol:
Hydroxyl group (
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- What is the correct assignment of the names of the functional groups in the following nitrogen- containing compounds? H3C 00 NCH2CH3 NOH 1 CH3 H3C NN(CH3)2 CH3 H3C CH3 3 2 1 = 2° amine; 2 = oxime; 3 = hydrazine 1 = imine; 2 = oxime; 3 = hydrazone = 1 amide; 2 = 2 amine; 3 = oxime 1 = imine; 2 = hydrazone; 3 = 2° aminearrow_forwardA reducing sugar will undergo the Fehling reaction, which requires a (potential) free aldehyde group. Which of the disaccharides as shown are reducing and which are nonreducing?arrow_forward(a) Label all the O atoms that are part of a glycoside in rebaudioside A. Rebaudioside A, marketed under the trade name Truvia, is a sweet glycoside obtained from the stevia plant, which has been used for centuries in Paraguay to sweeten foods. (b) The alcohol or phenol formed from the hydrolysis of a glycoside is called an aglycon. What aglycon and monosaccharides are formed by the hydrolysis of rebaudioside A?arrow_forward
- Which of the following is least soluble in hexane? * COOH A^ А B с E D FM COOH COOH COOH COOH 1 p COOHarrow_forward(2) ¿Lebäi The name of compound in the following image according to * IUPAC nomenclature is CH3 Br CH CH3 CH CH3 2-methyl-2-bromo-3-pentene O 4-bromo-4-methyl-2-pentene Oarrow_forwardWhy do ketoses go dehydration reaction much faster than aldoses in Seliwanoff’s test when aldehyde reacts faster than ketoses? Kindly explain in a comprehensive manner.arrow_forward
- A reducing sugar will undergo the Fehling reaction, which requires a(potential) free aldehyde group. Which of the disaccharides shown inFigure are reducing and which are nonreducing?arrow_forwardFor a sugar to behave as a reducing sugar, it must have a free aldehyde group. Fructose is a ketose, yet it behaves like a reducing sugar. Explain.arrow_forwardWith the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forward
- In solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forwardwhat is/are the product of hydrolysis with KOH?arrow_forwardWhich of the following terms correctly describes the relationship between D-fructose and L-fructose? O epimers diastereomers O enantiomers O cis-trans isomersarrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





