
Concept explainers
Interpretation:
The alcohol synthesised formed
Concept Introduction:
Oxidation and Reduction: The several organic molecules oxidation is a process by which a carbon atom gains to more electronegative elements most commonly oxygen, the reduction process by which a carbon atom gains bonds to less electronegative elements.
Reduction Reaction: This process in which any substance atoms, ion or molecule gains one or more electrons it is called reduction.
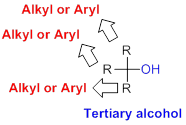
Tertiary alcohol:
Hydroxyl group (
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
- The following chemical reactants produce the ester ethyl ethanoate (C4H8O2): C2H6O + CH3COOH What type of reaction occurs to make ethyl ethanoate? condensation hydrolysis combustion acid-base reactionarrow_forwardA reducing sugar will undergo the Fehling reaction, which requires a(potential) free aldehyde group. Which of the disaccharides shown inFigure are reducing and which are nonreducing?arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forward
- Is galactose an aldehyde or a ketone sugar?arrow_forwardIn the monosaccharide derivatives known as sugar alcohols, the carbonyl oxygen is reduced to a hydroxyl group. For example, D-glyceraldehyde can be reduced to glycerol. However, this sugar alcohol is no longer designated D or L. Why?arrow_forwardWhat term best describes the isomeric relationship between alpha-D- glucopyranose and alpha-L-glucopyranose? A) structural isomers B) enantiomers C) epimers D) diastereoisomers, but not epimers E) not isomersarrow_forward
- Why might some food companies find iteconomically advantageous to advertise their product (for example, triacylglycerols) as being composed of polyunsaturated fattyacids with trans-double bonds?arrow_forwardWhat is the functional group that is usually associated with saponifiable lipid. Is it alcohol, ester, carboxylic acid or alkene?arrow_forwardThis is the compound tagatose. Is this an aldose or a ketose?arrow_forward
- Oxidation of a thiol produces a disulfide. True or Falsearrow_forwardWhy do ketoses go dehydration reaction much faster than aldoses in Seliwanoff’s test when aldehyde reacts faster than ketoses? Kindly explain in a comprehensive manner.arrow_forwardWhat is the carbohydrate classification for the product produced when each of the following undergoes complete hydrolysis?arrow_forward
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax

