
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
4th Edition
ISBN: 9780134465715
Author: John E. McMurry, David S. Ballantine
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 15.41AP
Draw the structures of the
(a) 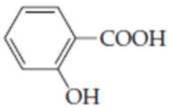
(b) 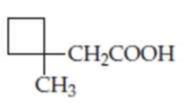
(c) CH3CH = CHCH2COOH
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?
Draw the full structure of triglyceride 1-docosahexanoyl-2-arachidonyl-3-elaidyl-glycerol with proper stereochemistry, given the following information:
docosahexanoic acid: 22:6n-3, arachidonic acid: 20:4n-6, elaidic acid: trans-18:1n-9,
Draw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
Ch. 15.1 - Prob. 15.1PCh. 15.1 - Prob. 15.2PCh. 15.2 - Draw structures corresponding to the following...Ch. 15.2 - Prob. 15.1CIAPCh. 15.2 - Prob. 15.2CIAPCh. 15.2 - Prob. 15.4PCh. 15.2 - Draw the line structures and provide common names...Ch. 15.2 - Prob. 15.6KCPCh. 15.3 - For each compound shown next (ad), indicate...Ch. 15.3 - Prob. 15.8KCP
Ch. 15.4 - Prob. 15.9KCPCh. 15.5 - Indicate whether the following compounds will give...Ch. 15.6 - Draw line structures of the following compounds...Ch. 15.6 - Prob. 15.12PCh. 15.7 - Prob. 15.13PCh. 15.7 - Prob. 15.14PCh. 15.7 - Prob. 15.15PCh. 15.7 - Draw the structure of each acetal or ketal final...Ch. 15.7 - Prob. 15.17PCh. 15.7 - Prob. 15.18PCh. 15.7 - Prob. 15.19PCh. 15.7 - Prob. 15.1MRPCh. 15.7 - Prob. 15.2MRPCh. 15.7 - Prob. 15.3MRPCh. 15.7 - Prob. 15.3CIAPCh. 15.7 - (a) What are mirotubules? (b) Why would drugs that...Ch. 15.7 - Tetrodotoxin, found in the puffer fish, has been...Ch. 15 - Prob. 15.20UKCCh. 15 - Prob. 15.21UKCCh. 15 - Prob. 15.22UKCCh. 15 - Prob. 15.23UKCCh. 15 - Prob. 15.24UKCCh. 15 - Prob. 15.25UKCCh. 15 - ALDEHYDESAND KETONES (SECTIONS 15.1 AND 15.2)...Ch. 15 - Draw a structure for a compound that meets each of...Ch. 15 - Prob. 15.28APCh. 15 - Prob. 15.29APCh. 15 - Draw structures corresponding to the following...Ch. 15 - Draw structures corresponding to the following...Ch. 15 - Prob. 15.32APCh. 15 - Prob. 15.33APCh. 15 - The following names are incorrect. What is wrong...Ch. 15 - Prob. 15.35APCh. 15 - Prob. 15.36APCh. 15 - Prob. 15.37APCh. 15 - Prob. 15.38APCh. 15 - Prob. 15.39APCh. 15 - Prob. 15.40APCh. 15 - Draw the structures of the aldehydes that might be...Ch. 15 - Write the structures of the hemiacetal or...Ch. 15 - Write the structures of the hemiacetal or...Ch. 15 - Prob. 15.44APCh. 15 - Prob. 15.45APCh. 15 - Prob. 15.46APCh. 15 - Prob. 15.47APCh. 15 - Prob. 15.48APCh. 15 - Prob. 15.49APCh. 15 - Prob. 15.50CPCh. 15 - Prob. 15.51CPCh. 15 - Prob. 15.52CPCh. 15 - Prob. 15.53CPCh. 15 - Prob. 15.54CPCh. 15 - Name the following compounds: (a) (b) (c) (be sure...Ch. 15 - Draw the structural formulas of the following...Ch. 15 - Prob. 15.57CPCh. 15 - Complete the following equations (refer to Summary...Ch. 15 - Prob. 15.59CPCh. 15 - How could you differentiate between 3-hexanol and...Ch. 15 - The liquids 1-butanol and butanal have similar...Ch. 15 - Prob. 15.62CPCh. 15 - Prob. 15.63GPCh. 15 - Prob. 15.64GPCh. 15 - Using the ketone structural form of fructose...Ch. 15 - Prob. 15.66GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw a Lewis structure for hydroxylamine, NH2OH.arrow_forwardThe reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardDraw condensed structural formulas for all products obtained from the complete hydrolysis of the following triacylglycerol.arrow_forward
- Draw the structure of a triacylglycerol containing stearic acid, palmitic acid, and oleic acid.arrow_forward1-34. Indicate whether each of the following statements about enantiomers is true or false. (a to d)arrow_forwardWrite the structure of L-Glutamic acid in pH 9 Write the Structure of L-Lysine in pH 1 2. Similarly write equations to show how L-lysine and L-Glutamic acid gives products when reacted with OH- ion as well as H3O+ ionarrow_forward
- Consider the intermolecular forces present in a pure sample of each of the following compounds: CH₃CH₂OH and CH₃COCH₃. Identify the intermolecular forces that these compounds have in common.arrow_forwardThe chemical formula for deoxyribose is C___ H___O____.arrow_forwardWrite the structural formula of a triacylglycerol that contains one unit each of lauric acid, palmitic acid, and oleic acid. How many other triacylglycerols, each containing all three of these acids, are possible?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Biodiversity hotspots and functional diversity; Author: Stockholm Resilience Centre TV;https://www.youtube.com/watch?v=Gr_eIsFOKr4;License: Standard Youtube License