
Concept explainers
(a)
Interpretation:
The product should be identified when the reaction of cyclopentanone react with given alcohol.
Concept Introduction:
A hemiacetal or a hemiketal: addition of alcohol to an
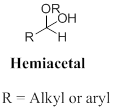
Ketal: A functional group bearing two alkyl group and two alkoxy groups are bonded to central carbon atom, the produced in the acid-catalysed alcoholysis (or) a hemiacetal. In other words addition of alcohol to the ketone or hemiacetal which produce ketal.
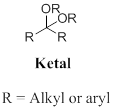
(b)
Interpretation:
The product should be identified when the reaction of cyclopentanone react with given alcohol.
Concept Introduction:
A hemiacetal or a hemiketal: addition of alcohol to an aldehyde or ketone which produce hemi acetal.
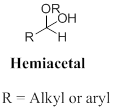
Ketal: A functional group bearing two alkyl group and two alkoxy groups are bonded to central carbon atom, the produced in the acid-catalysed alcoholysis (or) a hemiacetal. In other words addition of alcohol to the ketone or hemiacetal which produce ketal.
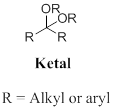
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
- The reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forwardTrehalose, a disaccharide found in the blood of insects, has the following structure. What simple sugars would you obtain on hydrolysis of trehalose?arrow_forwardAre the following molecules chiral or achiral? If they are chiral, identify the chiral carbon atom(s).(a) Pentan-3-ol (b) 2-Bromobutane(c) 2-Methylcyclohexanolarrow_forward
- For A, B, C, D, E, F, identify the circled functional groups and linkages in the compound in the picture.arrow_forwardFor the first part, draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. For the second part, identify whether the images are in D- or L-configuration. Write your answers first on a piece of bond paper.arrow_forwardAssume you could make a trisaccharide by covalently joining xylose (C5H10O5) , xylose (C5H10O5) and fructose (C6H12O6). So 2x xylose, 1x fructose. What would be the chemical formula of this trisaccharide assuming regular dehydration synthesis as discussed in class? You do not need to worry about which carbons are being used for forming these glycosidic linkages.arrow_forward
- Draw condensed structural formulas for all products obtained from the complete hydrolysis of the following triacylglycerol.arrow_forwardGlucose-1-phosphate has a ΔG°′ value of −20.9 kJ/mol, whereas that for glucose-6-phosphate is −12.5 kJ/mol. After reviewing the molecular structures of these compounds, explain why there is such a difference in these values.arrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. a) d- b) S- c) R- d) I-arrow_forward
- A reducing sugar will undergo the Fehling reaction, which requires a (potential) free aldehyde group. Which of the disaccharides as shown are reducing and which are nonreducing?arrow_forwardWith the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forwardThe 6-member pyranose ring of glucose is formed through the interaction of the hydroxyl group on C5 with the aldehyde group What kind of ring is formed when the hydroxyl group on C4 attacks instead? Draw the Haworth projection of the products with all the carbons labelled accordingly and name them Why would ring formation through C6 or C3 be unfavorable?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





