
(a)
Interpretation:
The compound that has higher solubility in water among the given pair has to be identified.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
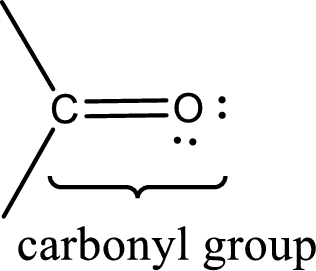
Aldehydes contain a carbonyl group that contains a hydrogen atom and a carbon atom bonded to it. Aldehyde that has one and two carbon atoms are gas at room temperature. The physical state of aldehyde that contains three carbon atoms to eleven carbon atoms that are not branched is liquid at room temperature. Aldehydes that contain more than eleven carbon atoms are solid at room temperature.
Ketones contain a carbonyl group that contains two carbon atoms bonded to it. For a compound to be ketone, a minimum of three carbon atom is required. Ketones that contain three carbon atoms to eight carbon atoms which have the carbonyl group at the second carbon atom are liquid at room temperature.
Solubility of aldehydes and ketones depend upon the length of the carbon chain. Those contain less than six carbon atoms are soluble in both water and organic solvents. Aldehydes and ketones that contain six or more carbon atoms are not soluble in water but soluble in organic solvents only. Solubility of smaller ketones and aldehydes are result of the hydrogen bond formation between the lone pairs of oxygen atom and the hydrogen atom in the water molecule.
(b)
Interpretation:
The compound that has higher solubility in water among the given pair has to be identified.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
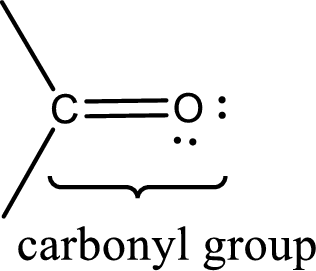
Aldehydes contain a carbonyl group that contains a hydrogen atom and a carbon atom bonded to it. Aldehyde that has one and two carbon atoms are gas at room temperature. The physical state of aldehyde that contains three carbon atoms to eleven carbon atoms that are not branched is liquid at room temperature. Aldehydes that contain more than eleven carbon atoms are solid at room temperature.
Ketones contain a carbonyl group that contains two carbon atoms bonded to it. For a compound to be ketone, a minimum of three carbon atom is required. Ketones that contain three carbon atoms to eight carbon atoms which have the carbonyl group at the second carbon atom are liquid at room temperature.
Solubility of aldehydes and ketones depend upon the length of the carbon chain. Those contain less than six carbon atoms are soluble in both water and organic solvents. Aldehydes and ketones that contain six or more carbon atoms are not soluble in water but soluble in organic solvents only. Solubility of smaller ketones and aldehydes are result of the hydrogen bond formation between the lone pairs of oxygen atom and the hydrogen atom in the water molecule.
Trending nowThis is a popular solution!

Chapter 15 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Match the chemical properties with the amino acid (note: the charges are not indicated on the charged amino acids) A. NH3+ - CH - COO- I (CH)2 I OH N,N - DimethyltryptamineAspartic acid B. NH3+ - CH - COO- I (CH)4 I NH2 Which one of each of these is: uncharged and polar charged and non-polar negatively charged and polar positively charged and polararrow_forwardDefine the following terms: a. hydrocarbon b. hydrophilic c. hydrophobic d. functional group e. R grouparrow_forwardFor each of the following chemicals, name the general class they belong to, discuss their solubility in water and explain why they are/are not soluble in water: a) CH3(CH2)26COOH b) KCl c) CH3arrow_forward
- The following is an example of a type of lipid. Answer the following questions based on its structure: OH a. What type of lipid is this categorized as: CH3 b. What two functional groups can be found in this structure? HO Estradiol (estrogen)arrow_forwardIn dehydration reaction between phosphate (derived from phosphoric acid) and glycerol (a sugar alcohol) accompanied with the removal of a water molecule, what chemical bond holds them together in a phospholipid molecule? Ether Bond Thioester Bond Acid Anhydride/phosphoanhydride bond Ester Bondarrow_forwardWhich of the following statements is correct regarding polar/nonpolar molecules? 1. Molecules comprised of nonpolar covalent bonds are likely to be hydrophilic 2. Polar molecules like sugar tend to dissolve in solutions of other polar molecules like water 3. Nonpolar molecules like sugar tend to dissolve in solutions of polar molecules like water 4. Polar covalent bonds tend to form between molecules that consist of two atoms of the same element, like oxygen gas (O2) or nitrogen gas (N2)arrow_forward
- When a water droplet is placed in an oil solution, the phospholipids will form a micelle as shown. Explain why the phospholipids become organized in this way. Oil Waterarrow_forward70 degree Celcious is the melting point of stearic acid, and 4 degree Celcius is the melting point of oleic acid. Explain using the concept of lipids why their melting point is so different.arrow_forwardLook at the structure of stearic acid as well as oleic acid and answer the following questions: a.Write which of these two is saturated fatty acid. b.Which of these two has a higher melting points. c.Which of these two will change the color of bromine water to clear. d.Name the unsaturated fatty acid using both delta and Omega nomenclature I can not separate the question, it is one question with four parts. I only need "D" answered, pleasearrow_forward
- When a hydrophobic substance like a hydrocarbon is dissolved in water, a clathrate cage of ordered water molecules is formed about it. If we consider only the effects on water, what do you expect the sign of ∆S to be for this process? Explain your answer.arrow_forwardWhich of the following statements is/are true? 1. Sphingolipids all have a palmitate derivative that is ester-linked to the sphingosine backbone. 2. The addition of cholesterol will always lead to an increase in the rigidity of a lipid bilayer. 3. Glycolipids may have a sphingosine or a glycerol backbone to which a sugar moiety is attached. 4. All lipid molecules are characterized by the presence of a carboxylic acid functional group. O 1 and 2 O 1 only O 2 and 3 O 3 only O 1,3, and 4arrow_forward_______ groups are the acid part of amino acids and fatty acids. a. Hydroxyl (OH) c. Methyl (CH3) b. Carboxyl (COOH) d. Phosphate (PO4)arrow_forward
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

