
Interpretation:
To give the explanation whether the acid-catalyzed hydrolysis of acetamide is a reversible or irreversible reaction.
Concept introduction:
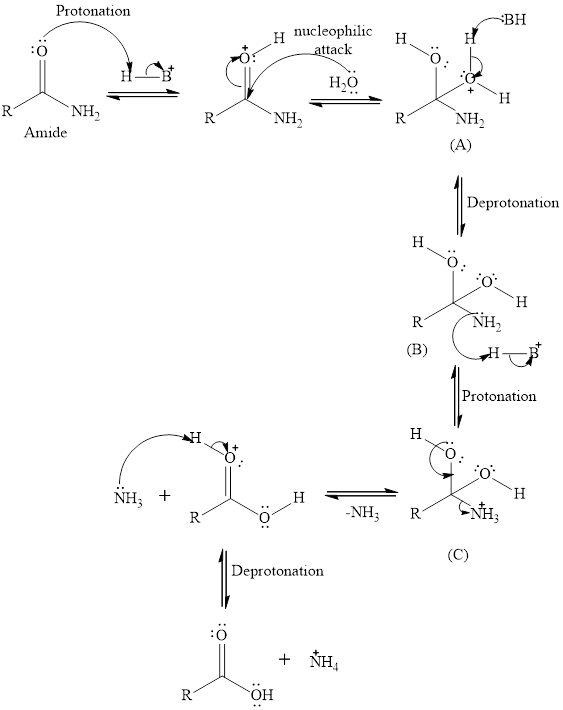
In acid hydrolysis of amide, acid protonates carbonyl oxygen of amide which increases the electrophilicity of the carbonyl carbon. Water acts as a nucleophile gets attached to the carbonyl carbon of amide to form tetrahedral intermediate(A) which on losing proton get converted to tetrahedral intermediate(B). The nitrogen of tetrahedral intermediate (B) gets protonated to form tetrahedral intermediate(C). The
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
- Choose among these options Among these Inhibits the enzyme which is responsible for the assembly of matured viral particles Saquinavir Didanosine Penciclovir Azidothymidine The following are essential components in the structure of sulfonamides with the exception of Amino group Benzene ring Pyridine N-Acyl amino This drug has been associated with optic neuritis and therefore discontinued after 2 months of treatment Isonizaid Rifampicin Ethambutol Pyrazinamide A non prodrug ACE Inhibitor Captopril Enalapril Quinapril Fosinopril High sodium diet High fat diet Low potassium diet Low phosphate dietarrow_forwardSome penicillins cannot be administered orally because their β-lactam is rapidly hydrolyzed by the acidic environment of the stomach. What product is formed in the following hydrolysis reaction?arrow_forwardWhat is the difference between the aminolysis of an ester and that of an acyl halide?arrow_forward
- What is the Methods used to Synthesis of IMINES.arrow_forwardBisphenol A is widely used as a building block in polymer synthesis and is found in the polycarbonate hard plastics of reusable drink containers, DVDs, cell phones, and other consumer goods. Bisphenol A is reported to have estrogenic activity, and its widespread occurrence in our environment is a potential concern. Describe one or two biochemical experiments that could be done to compare the activity of bisphenol A with that of its estradiol, its structural relative.arrow_forwardClick the "draw structure" button to activate the drawing utility. Some penicillins cannot be administered orally because their B-lactam is rapidly hydrolyzed by the acidic environment of the stomach. Draw the product that forms in the following hydrolysis reaction. H H R. H3O+ -N- но HINarrow_forward
- Describe the Concept of Acid-Catalyzed Ester Hydrolysis and Transesterification.arrow_forwardCardiolipins are found in heart muscles. Draw the products formed when a cardiolipin undergoes complete acid-catalyzed hydrolysis.arrow_forwardWhat are the physicochemical properties of ISOAMYL ACETATE?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole





