
(a)
Interpretation:
To propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride and how acetic anhydride makes it easier to form the succinic anhydride.
Concept introduction:
Succinic anhydride is a type of cyclic anhydride formed by the dehydration of dicarboxylic acid that is succinic acid. The cyclic anhydride is formed easily in the presence of anhydride such as acetic anhydride.
The reaction is:
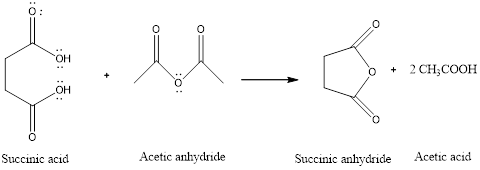
(b)
Interpretation:
To propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride and how acetic anhydride makes it easier to form the succinic anhydride.
Concept introduction:
Succinic anhydride is a type of cyclic anhydride formed by the dehydration of dicarboxylic acid that is succinic acid. The cyclic anhydride is formed easily in the presence of anhydride such as acetic anhydride.
The reaction is:

Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
- By means of a suitable reaction, show how each of the compounds can be prepared from propionic acid. More than one step may be required. a. methyl propylamine (CH3NHCH2CH2CH3)b. propionyl chloridec. ethyl propionated. propionic anhydridee. N-methyl propionamidearrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Why does the Ritter reaction not work with primary alcohols? b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.arrow_forwardNabumetone is a pain reliever and anti-inflammatory agent sold under the brand name of Relafen.a. Write out a synthesis of nabumetone from ethyl acetoacetate.b. What ketone and alkyl halide are needed to synthesize nabumetone by direct enolate alkylation?arrow_forward
- 2. Explain why the acetylation requires one mole of acetic anhydride per mole of aniline, though acetic anhydride contains two acetyl groups. How is the by-product containing the second acetyl group removed from acetanilide?arrow_forwardHow could you convert N-methylbenzamide to the following compounds? a. N-methylbenzylamine b. benzoic acid c. methyl benzoate d. benzyl alcoholarrow_forwarda. What two amides are obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of propylamine?b. Why is only one amide obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of triethylamine?arrow_forward
- The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Propose a mechanism for the Ritter reaction.b. Why does the Ritter reaction not work with primary alcohols?c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?arrow_forward1. Show how to synthesis the following compounds using either a malonic ester synthesis or an acetoacetic ester synthesis? 2-Butylhexanoic acid b. Cyclohexyl methyl ketone a.arrow_forwardNabumetone is a pain reliever and anti-inammatory agent sold under the brand name of Relafen.a. Write out a synthesis of nabumetone from ethyl acetoacetate.b. What ketone and alkyl halide are needed to synthesize nabumetone by direct enolate alkylation?arrow_forward
- 12. You can distinguish epinephrine hydrotartrate from norepinephrine hydrotartrate by? A. Water solubility B. Reactions of oxidation with iodine at different pH C. Reactions in general alkaloid precipitation reagents D. Reactions with iron(III) chloride E. Reactions in Fehling's reagent.arrow_forward1. What are the possible interferences or complications in detecting and differentiating aldehydesand ketones using the following tests:a. 2,4-dinitrophenyl hydrazine test b. benedicts test c. tollens test d. jones test e. iodoform test 2. Describe the positive results for the following qualitative tests:a. 2,4-dinitrophenyl hydrazine testarrow_forwardWhat are the products of the following reactions? (A trace amount of acid is present in each case.) a. cyclopentanone + ethylamine c. acetophenone + hexylamine b. cyclopentanone + diethylamine d. acetophenone + cyclohexylaminearrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

