
Concept explainers
Catalytic antibodies catalyze a reaction by forcing the conformation of the substrate in the direction of the transition state. The synthesis of the antibody is carried out in the presence of a transition state analog—a stable molecule that structurally resembles the transition state. This causes an antibody to be generated that recognizes and binds to the transition state, thereby stabilizing it. For example, the following transition state analog has been used to generate a catalytic antibody that catalyzes the hydrolysis of the structurally similar ester:
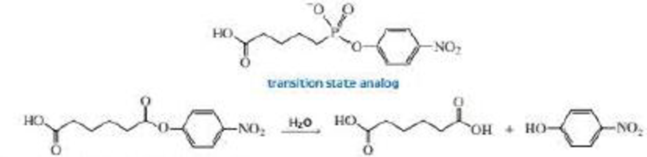
a. Draw a possible transition state for the hydrolysis reaction.
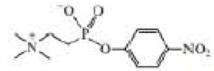
b. The following transition state analog is used to generate a catalytic antibody for the catalysis of ester hydrolysis. Draw the structure of an ester whose rate of hydrolysis would he increased by this catalytic antibody.
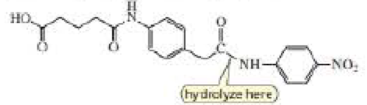
c. Design a transition state analog that catalyzes amide hydrolysis at the amide group indicated.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
- In the "Catalysis by Preferential Transition State Binding" mechanism, the substrate perfectly fits with the enzyme binding site after its geometry gets changed. Who does it? And how?arrow_forwardChemistry Calculate the substrate concentration of an enzyme catalyzed reaction whose rate is equal to half of its maximum velocity. Also evaluate Michaelis Menten constant and substrate concentration when the rate is equal to its maximum velocity.arrow_forwardSelect below all the catalytic mechanisms used by chymotrypsin to convert substrate into product. metal ion catalysis covalent catalysis acid-base catalysisarrow_forward
- What kind of mechanistic step is represented in the following process? elimination termination initiation O propagation minarrow_forwardWhat do you predict the addition of an acid will have on catalase? Will it increase, decrease, or have no effect on the chemical reaction between hydrogen peroxide and catalase?arrow_forwardSelect the intermediate species that does NOT participate in the mechanism of this reaction;arrow_forward
- Consider the simple reaction S <--> P, with the forward rate characterized by a rate constant k1, the reverse rate constant characterized by a rate constant k2, and the equilibrium constant for the reaction given by Keq. Which of the listed effects would be brought about by an enzyme catalyzing this reaction?a) increased Keqb) increased k1c) decreased transition state free energy changed) increased k2arrow_forwardWhat do you predict the addition of a base will have on catalase? Will it increase, decrease, or have no effect on the chemical reaction between hydrogen peroxide and catalase?arrow_forwardwhat is the approximation used to derive the reaction rate for an enzyme catalysed reaction that proceeds by a michaelis menten mechansimarrow_forward
- Juan performed the same chemical reaction twice. His data from the first at- tempt is indicated by the red line. His data from the second attempt is indicated by the blue line. Energy During a Chemical Reaction Energy A B reactants e.g. C6H12O6 + O₂ C without enzyme Juan added pressure. with enzyme Reaction coordinate What change did Juan most likely make to his reaction the second time he per- formed it? activation energy with enzyme D Juan added a catalyst. Juan increased the surface area of the reactants. Juan cooled his reactants. activation energy without enzyme overall energy released during reaction products CO,+H,Oarrow_forwardWhich of the following factors does not influence the rate of an enzyme catalyzed reaction?arrow_forwardAspirin containing acetylsalicylic acid is used, dichloromethane is added, subsequently sodium hydroxide is added and hydrochloric acid is finally added, which is the reaction mechanism and if acetylsalicylic acid is formed at the end of the entire reaction.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


