
Concept explainers
Catalytic antibodies catalyze a reaction by forcing the conformation of the substrate in the direction of the transition state. The synthesis of the antibody is carried out in the presence of a transition state analog—a stable molecule that structurally resembles the transition state. This causes an antibody to be generated that recognizes and binds to the transition state, thereby stabilizing it. For example, the following transition state analog has been used to generate a catalytic antibody that catalyzes the hydrolysis of the structurally similar ester:
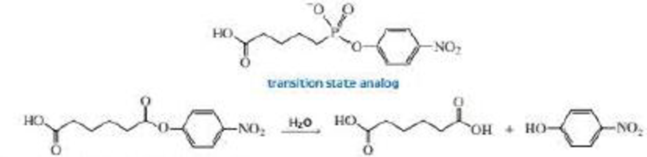
a. Draw a possible transition state for the hydrolysis reaction.
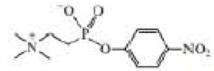
b. The following transition state analog is used to generate a catalytic antibody for the catalysis of ester hydrolysis. Draw the structure of an ester whose rate of hydrolysis would he increased by this catalytic antibody.
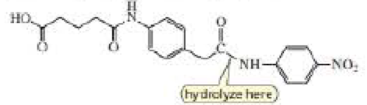
c. Design a transition state analog that catalyzes amide hydrolysis at the amide group indicated.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry - MasteringChemistry
- subunits D,E,F help pleasearrow_forwardEnzyme Catalysis: Make a generalizations about the experiment.arrow_forwardAmylase’ is an enzyme in our saliva that digests the ‘amylose’ protein in mouth. How could you justify the mechanism of enzymatic action using enzyme-catalyzed reaction models?arrow_forward
- Aspirin containing acetylsalicylic acid is used, dichloromethane is added, subsequently sodium hydroxide is added and hydrochloric acid is finally added, which is the reaction mechanism and if acetylsalicylic acid is formed at the end of the entire reaction.arrow_forwardafter you write the five reacitons pathways, label the rate-determining step and mechanism pleasearrow_forwardI drew out the reaction and the transition states but I am not sure how to draw the energy diagram for compounds with similar mechanismsarrow_forward
- H2 + CH2 = CH2 --> CH3CH3Which of the following could be a substrate for an enzyme that has an induced-fit active site and catalyzes the reaction above? a. CH3_OH b. CH2=CH_CH3 c. CH3_O_CH2CH3 d. none of the abovearrow_forwardCarbonic anhydrase facilitates the dissolution of carbon dioxide gas into water by catalyzing its hydration. Is the catalyzed reaction efficient? By what criterion do you state that?arrow_forwardPlease answer with the basic mechanism ASAParrow_forward

 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


