
Concept explainers
(a)
Interpretation:
To predict the products of the following transesterification reaction.
(a)
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
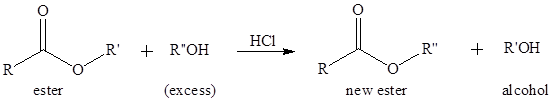
(b)
Interpretation:
To predict the products of the following transesterification reaction.
(b)
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
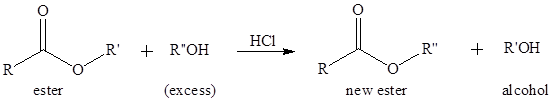
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry - MasteringChemistry
- a. Propose a mechanism for the reaction of acetic anhydride with water.b. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?arrow_forwardWhat is the best set of reagents to use for the synthesis?arrow_forwardExplain Reaction of Grignard Reagents with Carbon Dioxide ?arrow_forward
- Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidarrow_forwardwhat would the best reagents be? And why?arrow_forwardThe name for Reaction 1 is _____ while Reaction 2 is called _____. Choices: A. Williamson Ether synthesis, B. Hydration, C. Epoxidation, D. Acidic cleavage The reagent/s for REACTION 1 is/are ______ Choices: A. m-chloroperoxybenzoic acid, B. H2O/H3PO4, C. H3O+, D. HI The reagent/s for REACTION 2 is/are ______ Choices: A. m-chloroperoxybenzoic acid, B. water in acidic medium, C. dilute acid, D. hydroiodic acidarrow_forward
- Explain the Reaction of Organometallic Reagents with Epoxides ?arrow_forwardLinalool (the Chapter 9 opening molecule) and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can’t linalool be prepared by a similar pathway?arrow_forward1. i)Give the structure of the product from the reaction of propanal with 1M ethanol in dry acid . ( II)What happens when further 1M of ethanol is added to the abovearrow_forward
- Which reagents would you use to perform reaction B?a) PBr3.b) p-toluenesulfonyl chloride.c) SOCl2.d) NaOCl, NaOHarrow_forward(a) How will you carry out the following conversions?(i) Acetylene to Acetic acid (ii) Toluene to m-nitrobenzoic acid(iii) Ethanol to Acetone(b) Give reasons :(i) Chloroacetic acid is stronger than acetic acid.(ii) pH of reaction should be carefully controlled while preparing ammonia derivatives of carbonyl compounds.arrow_forwardA greener alternative to bromination with elemental bromine is the reaction of the acetanilide with potassium bromide and ammonium ceric nitrate. How does this reaction work?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


