
(a)
Interpretation:
To propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride and how acetic anhydride makes it easier to form the succinic anhydride.
Concept introduction:
Succinic anhydride is a type of cyclic anhydride formed by the dehydration of dicarboxylic acid that is succinic acid. The cyclic anhydride is formed easily in the presence of anhydride such as acetic anhydride.
The reaction is:
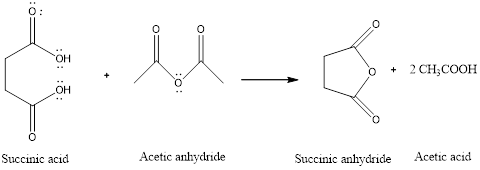
(b)
Interpretation:
To propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride and how acetic anhydride makes it easier to form the succinic anhydride.
Concept introduction:
Succinic anhydride is a type of cyclic anhydride formed by the dehydration of dicarboxylic acid that is succinic acid. The cyclic anhydride is formed easily in the presence of anhydride such as acetic anhydride.
The reaction is:

Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry - MasteringChemistry
- Design a stepwise synthesis to convert cyclopentanone and 4-bromobutanal to hydroxy aldehyde A.arrow_forwardplease explain why phenol is not easily attacked by a positive reagent such as aniline?arrow_forwardwhy sulfanilamide is less acidic than sulfathiazole (B), whereas sulfanilylsulfanilamide is more acidic than sulfathiazole? does it relate to hard and soft nucleophiles?arrow_forward
- Why is an alkylamine more basic than ammononia?arrow_forwarda. Propose a mechanism for the reaction of acetic anhydride with water.b. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?arrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Why does the Ritter reaction not work with primary alcohols? b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.arrow_forward
- Show the reagent needed to convert butanoic acid to its derivative ofi. butanoyl chlorideii. butanamideiii. butanoliv. methyl butanoatearrow_forwardWhy do we not isolate any ortho-nitrated product? (In a Acetanilide Nitration)arrow_forwardWhy can you not use this reaction to make a secondary amine? a) Alkyl halides are weak electrophiles.b) Primary amines are weak nucleophiles.c) The primary amine is too nucleophilic, so over-reaction will occur.d) The secondary amine product is more nucleophilic than the primary amine reactant, so over-reaction will occur.arrow_forward
- 4-methoxybenzoic acid is less or more polar than 4-methoxyacetophenone? explain why (WITHOUT DRAWINGS)arrow_forwardWhats the reason for the girgnard reagent attack that carbon of starting point in this step? a. Bc it gives us desired product b. Due to the sterics c. Bc it is more electrophilic than the other carbon of the starting pointarrow_forwardHow do you explain the difference in colors between the hydrazone product formed from simple aldehyde/ketone and aromatic aldehyde/ketone (yellow vs orange) in Dinitrophenylhydrazine Test?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
