
Interpretation:
To predict that out of alcohol, water, and
Concept introduction:
Acid anhydride can react with water, alcohol and amine. When acid anhydride reacts with water it gives two equivalent of carboxylic acid, with alcohol it gives carboxylic acid and ester, with amine it gives carboxylate ion and amide. Amine water and alcohol acts as a nucleophile and gets attached to the carbonyl carbon of the acid anhydride. The base takes the proton from the from the tetrahedral intermediate. The carboxylate ion gets eliminated from the tetrahedral intermediate to form carboxylic acid derivative. The mechanism of this reaction is:
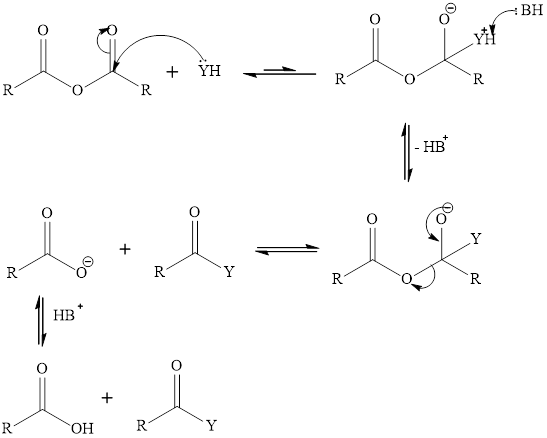
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry - MasteringChemistry
- The structure below represents a tetrahedral alkoxide-ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Draw both the starting acid derivative and the ultimate carboxylic acid derivative product. Do not include the product formed by the leaving group.arrow_forwardWhat type of eliminaton reactions occur in this process?arrow_forwardThe hydrolysis of the ester shown here is catalyzed by morpholine. Explain how morpholine catalyzes the reaction. (Hint: The pKa of the conjugateacid of morpholine is 9.3, so morpholine is too weak a base to function as a base catalyst.)arrow_forward
- Given the information pictured, The final step of the reaction sequence uses PhLi. Provide a reason and possible mechanism by which this final elimination and hydrolysis reaction would occur.arrow_forwardComplete the following sequence of reactions, giving structural details of all key intermediatesarrow_forwardDescribe the following giving the relevant chemical equation in each case: (i) Carbylamine reaction (ii) Hoffmann’s bromamide reaction.arrow_forward
- please explain why this happends, im having trouble in this section. Part A Chymotrypsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the small intestine at an optimum pH of 7.7 to 8.0. How is the rate of the chymotrypsin-catalyzed reaction affected by each of the following conditions?arrow_forwardThe trityl cation intermediate is a highly stable species due to the resonance stabilization provided by the phenyl rings. Please draw these resonance structures.arrow_forward22.26 In each of the following reactions, the aromatic ring has just one chemically distinct, aromatic H, so a single electrophilic aromatic substitution will lead to just a single product. With this in mind, predict the product of each of these reactions (b) NO2 (c) Br Chn FeCl3 Cl AlCl AIC NO2arrow_forward
- Which of the following is NOT a catalytic strategy used by carbonic anhydrases? a. metal ion catalysis b. catalysis by approximation c. Acid-base catalysis d. Formation of covalent intermediatearrow_forwardIn the mechanism involving the dehydration of an alcohol, which alkoxonium ion has the highest amount of energy? A. secondary alkoxonium ion B. methyloxonium ion C. tertiary alkoxonium ion D. primary alkoxonium ionarrow_forwardWrite the mechanism for the following reactions. Explain detail each steps of reactions.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


