
Concept explainers
(a)
Interpretation:
Given the each set of compounds whether it is a hemiacetal (or) neither should be identified.
Concept Introduction:
A hemiacetal or a hemiketal: addition of alcohol to an
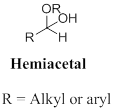
(b)
Interpretation:
Given the each set of compounds whether it is a acetal, ketal (or) neither should be identified.
Concept Introduction:
Acetals: Generally Acetals are used to protect the aldehyde (carbonyl group).
In this reaction acetaldehyde is protected as acetal by using methanol.
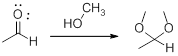
A hemiacetal or a hemiketal: addition of alcohol to an aldehyde or ketone which produce hemi acetal.
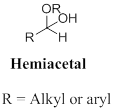
(c)
Interpretation:
Given the each set of compounds whether it is a acetal, ketal (or) neither should be identified.
Concept Introduction:
A hemiacetal or a hemiketal: addition of alcohol to an aldehyde or ketone which produce hemi acetal.
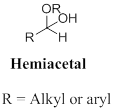
Ketal: A functional group of two alkyl group and two alkoxy groups are bonded to central carbon atom, the produced by the acid-catalysed alcoholysis (or) a hemiacetal. In other words addition of alcohol to the ketone or hemiacetal which produce ketal.
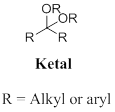
Cyclic hemiacetals: Intramolecular hydroxyl group reacts with the carbonyl group which forms cyclic hemiacetals.
(d)
Interpretation:
Given the each set of compounds whether it is a hemiacetal, hemiketal (or) neither should be identified.
Concept Introduction:
Ketal: A functional group bearing two alkyl group and two alkoxy groups are bonded to central carbon atom, it produced in the acid-catalysed alcoholysis (or) a hemiacetal.
Hemiketals: A compound with both an alcohol (-OH) groups and an ether (-OR) groups are bonded to the carbon atom that was at one time the ketone carbonyl carbon.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- What is the structural difference between a hemiacetal and an acetal?arrow_forwardWhat structural feature is necessary for an alcohol to undergo oxidation reactions?arrow_forwardA 5 ml vial of the hydrocortisone is having 2% (w/v) concentration. How many vials will be required, if we needed to prepare 30 ml of a 8 mg/ml oral hydrocortisone?arrow_forward
- The chemical formula for deoxyribose is C___ H___O____.arrow_forwardA 5 ml vial of hydrocortisone is having 2% (w/v) concentration. How many vials will be required, if we need to prepare 30 ml of a 8 mg/ml oral hydrocortisone?arrow_forwardWhat is the composition of the soap formed by basic hydrolysis of each triacylglycerol?arrow_forward
- Trehalose, a disaccharide found in the blood of insects, has the following structure. What simple sugars would you obtain on hydrolysis of trehalose?arrow_forwardFor the first part, draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. For the second part, identify whether the images are in D- or L-configuration. Write your answers first on a piece of bond paper.arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forward
