
Concept explainers
(a)
Interpretation:
Structural formula for methyl thiopropanoate has to be drawn.
Concept Introduction:
General structure of thioester can be represented as shown below,
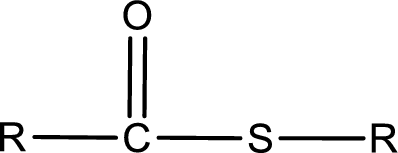
From an IUPAC name, the structure of the thioester can be derived. IUPAC name of thioester consists of two parts. In an IUPAC name of thioester, the first part of the name is the alkyl part and the second part is the acid part. Alkyl group have come from the thiol and acyl part from
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of thiocarboxylic acid.
(b)
Interpretation:
Structural formula for methyl thiopropionate has to be drawn.
Concept Introduction:
General structure of thioester can be represented as shown below,
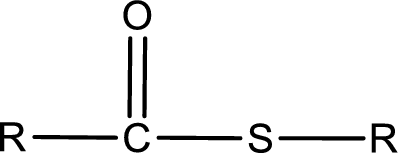
From an IUPAC name, the structure of the thioester can be derived. IUPAC name of thioester consists of two parts. In an IUPAC name of thioester, the first part of the name is the alkyl part and the second part is the acid part. Alkyl group have come from the thiol and acyl part from carboxylic acid.
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of thiocarboxylic acid.
(c)
Interpretation:
Structural formula for ethyl thioacetate has to be drawn.
Concept Introduction:
General structure of thioester can be represented as shown below,
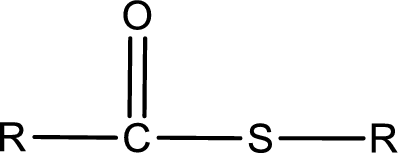
From an IUPAC name, the structure of the thioester can be derived. IUPAC name of thioester consists of two parts. In an IUPAC name of thioester, the first part of the name is the alkyl part and the second part is the acid part. Alkyl group have come from the thiol and acyl part from carboxylic acid.
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of thiocarboxylic acid.
(d)
Interpretation:
Structural formula for ethyl thioethanoate has to be drawn.
Concept Introduction:
General structure of thioester can be represented as shown below,
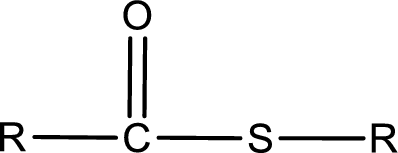
From an IUPAC name, the structure of the thioester can be derived. IUPAC name of thioester consists of two parts. In an IUPAC name of thioester, the first part of the name is the alkyl part and the second part is the acid part. Alkyl group have come from the thiol and acyl part from carboxylic acid.
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of thiocarboxylic acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
GENERAL,ORGANIC,+BIO.CHEM.-MINDTAP
- Which of the following organic compounds has a functional group of a carboxyl group?A. alkyl halide B. ester C. ketone D. aminearrow_forwardWhat kind of solvent ingredients is widely is usually used in mouthwash, perfumes and spray A. Ethly acetate B. Aliphatic alcohols C. Fragrant esters D. Phenols and phenol derivativesarrow_forwardWhat is hydrolysis of ethyl acetate? What are those example of it.arrow_forward
- What are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. acetic acid and toluene b. phenylic acid and ethanol C. ethanoic acid and benzene d. benzoic acid and ethanol e. phenylic acid and methanol O a O barrow_forwardWhat are the major products of the reaction of ethyl benzoate with hydrochloric acid and water? a. benzoic acid and ethanol b. phenylic acid and ethanol c. ethanoic acid and benzene d. acetic acid and toluene e. phenylic acid and methanolarrow_forward1. Draw the structural formula of the hemiacetal formed from each ơf the following pairs of reactants. a. Acetaldehyde and methanol 0 b. 2-Pentanone and ethyl alcohol С. Butanal and isopropyl alcohol d. Acetone and ethanolarrow_forward
- Formation of Esters. Odor of ester Chemical Equation:arrow_forwardThis carboxylic salts are effective ingredient against yeast and molds in beverages, jams, pie fillings and ketchup a. benzoates b. sorbates c. acetates d. propionatesarrow_forward1. Which of the following structural features is possessed by aldehydes but not ketones? a. At least one hydroxyl group is bonded to the carbonyl carbon atom. b. At least one hydrogen atom is bonded to the carbonyl carbon atom. c. The carbonyl carbon atom is bonded to two other carbon atoms. d. The carbonyl carbon atom is part of a ring structure. 2. What is the IUPAC name of the following compound in the attached photo? a. 3-chloro-4-nitrobenzoic acid b. 2-chloro-4-carboxynitrobenzene c. 2-nitro-5-carboxychlorobenzene d. 2-chloro-1-nitro-4-benzoic acid 3. Which of the following is the correct bond-line structure for CH3C≡C(CH2)2CH(CH3)2? a. Structure I b. Structure II c. Structure III d. Structure IVarrow_forward
- Write the condensed formula for each of the following compounds.a. 3-Methylnonanal c. 4-Fluorohexanalb. b-Bromovaleraldehyde d. a,b-Dimethylbutyraldehydearrow_forward. The following chemical reactants produce the ester ethylethanoate (C4H8O2):C2H6O + CH3COOHWhat type of reaction occurs to make ethyl ethanoate?a. condensationb. hydrolysisc. combustiond. acid-base reactionarrow_forwardMultple choice 4. To which family of organic compounds does CH₂CH₂CH₂CH₂CHO a. Alcohol b. Aldehyde c. Alkyne 5. Which statement is correct? belong? d. Ketone e. Carboxylic acid a. Aldehydes strongly resist oxidation. b. Ketones are easily oxidized. c. Carboxylic acid groups are easily oxidized. d. An ester may be hydrolyzed to give a carboxylic acid and an alcohol. e. Amides will not react with water. 6. Which statement describes addition polymers? a. They are made of monomers that have two functional groups. b. They may form between an organic acid and an amine. c. When they are produced, a small molecule (such as water) is also produced. d. all of the above e. None of the abovearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





