
Concept explainers
(a)
Interpretation:
Lactic acid has to be classified among the given options.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
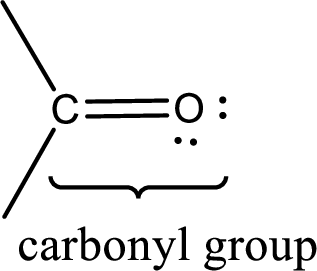
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
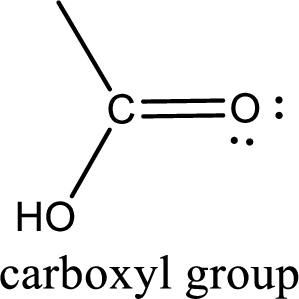
Apart from carboxyl group, there can be other functional group also that is present in the
(b)
Interpretation:
Glutaric acid has to be classified among the given options.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
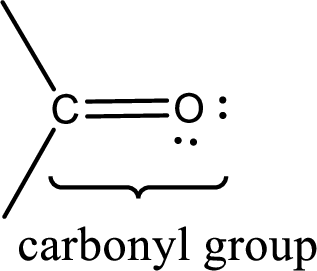
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
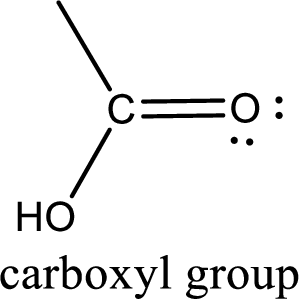
Apart from carboxyl group, there can be other functional group also that is present in the carboxylic acid. They are known as polyfunctional carboxylic acids. These occur naturally also. Some of the important type of polyfunctional carboxylic acids are hydroxy acids, keto acids, and unsaturated acids.
(c)
Interpretation:
Valeric acid has to be classified among the given options.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
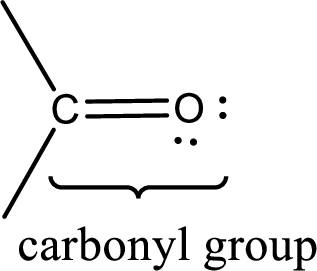
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
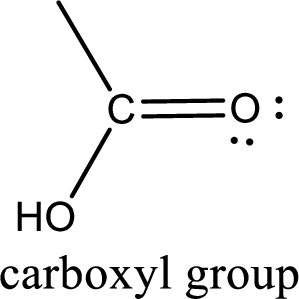
Apart from carboxyl group, there can be other functional group also that is present in the carboxylic acid. They are known as polyfunctional carboxylic acids. These occur naturally also. Some of the important type of polyfunctional carboxylic acids are hydroxy acids, keto acids, and unsaturated acids.
(d)
Interpretation:
Pyruvic acid has to be classified among the given options.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
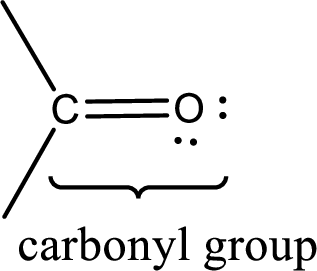
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
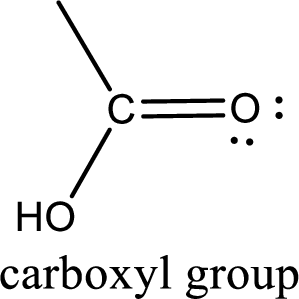
Apart from carboxyl group, there can be other functional group also that is present in the carboxylic acid. They are known as polyfunctional carboxylic acids. These occur naturally also. Some of the important type of polyfunctional carboxylic acids are hydroxy acids, keto acids, and unsaturated acids.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
GENERAL,ORGANIC,+BIO.CHEM.-MINDTAP
- Classify the two carboxylic acids in each of the following pairs as (A) both dicarboxylic acids, (B) both monocarboxylic acids, or (C) one dicarboxylic and one monocarboxylic acid. 1. Glutaric acid and valeric acid2. Adipic acid and oxalic acid3. Caproic acid and formic acid4. Succinic acid and malonic acidarrow_forwardGive an example of a medical application of aspirin. Why was aspirin modified and patented into its ester acetylsalicylic acid form that we know today?arrow_forwardComplete the table provided below for nomenclature of Carboxylic Acid & Derivatives.arrow_forward
- Carboxylic Acid Alcohol Ester (Condensed Structural Formula) Odo Formic acid Ethyl alcohol Rum Acetic acid Ethyl alcohol Fruity Acetic acid Isopentyl (isoamyl) alcohol Banana Acetic acid Octyl alcohol Orange Propionic acid Ethyl alcohol Pineapplearrow_forwardWhat is porphyrin ?arrow_forwardHow Chemists Activate Carboxylic Acids.arrow_forward
- 1) To identify which contains more acid vinegar or lemon juice neutralization with sodium hydroxide can be used. Explain how. 2) The long filaments making up the thread of woven synthetic fabrics are likely polyesters. Describe the special characteristic that the alcohol and carboxylic acid must have so that they can react with each other to produce such long filaments.arrow_forwardFormation of Esters. Odor of ester Chemical Equation:arrow_forwardwhy n-hexanol has higher boiling point than n-hexane. why acetic acid is more acidic then butanoic acid. why sugar is soluble in water in terms of their molecular interactions.arrow_forward
- 4. Draw the chemical structure of acetic anhydride and acetyl salicylic acid.arrow_forwardEach of the following acids contains an additional type of functional group besides the carboxyl group. For each acid, specify the non-carboxyl functional group present. 12. Glycolic acid13. Fumaric acid14. Pyruvic acid15. Malic acid16. Tartaric acidarrow_forwardWhat is the units of the following constant: a Acid valueb. Saponification valuec. Ester valued. Unsaponifiable mattere. Iodine valuef. Hydroxyl valueg. Acetyl value of fatty acidsarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



