
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN: 9781305580343
Author: Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 16.32QP
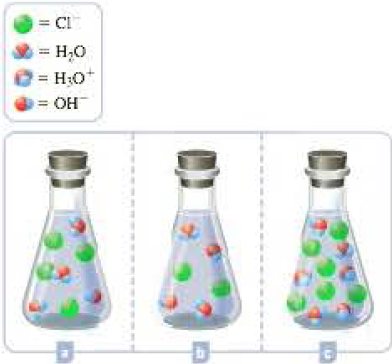
The three flasks shown below depict the titration of an aqueous NaOH solution with HCl at different points. One represents the titration prior to the equivalence point, another represents the titration at the equivalence point, and the other represents the titration past the equivalence point. (Sodium ions and solvent water molecules have been omitted for clarity.)
- a Write the balanced chemical equation for the titration.
- b Label each of the beakers shown to indicate which point in the titration they represent.
- c For each solution, indicate whether you expect it to be acidic, basic, or neutral.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 16 Solutions
General Chemistry - Standalone book (MindTap Course List)
Ch. 16.1 - Lactic acid, HC3H5O3, is found in sour milk, where...Ch. 16.1 - What are the concentrations of hydrogen ion and...Ch. 16.1 - What is the pH of an aqueous solution that is...Ch. 16.1 - You have prepared dilute solutions of equal molar...Ch. 16.2 - Sulfurous acid, H2SO3, is a diprotic acid with Ka1...Ch. 16.3 - Quinine is an alkaloid, or naturally occurring...Ch. 16.3 - What is the hydronium-ion concentration of a 0.20...Ch. 16.3 - Prob. 16.2CCCh. 16.4 - Consider solutions of the following salts: a....Ch. 16.4 - Calculate the following at 25C, using Tables 16.1...
Ch. 16.4 - Benzoic acid, HC7H5O2, and its salts are used as...Ch. 16.4 - Which of the following aqueous solutions has the...Ch. 16.5 - The chemical equation for the hydrolysis of...Ch. 16.5 - What is the concentration of formate ion, CHO2, in...Ch. 16.5 - One liter of solution was prepared by dissolving...Ch. 16.6 - What is the pH of a buffer prepared by adding 30.0...Ch. 16.6 - Suppose you add 50.0 mL of 0.10 M sodium hydroxide...Ch. 16.6 - Prob. 16.5CCCh. 16.6 - The beaker on the left below represents a buffer...Ch. 16.7 - What is the pH of a solution in which 15 mL of...Ch. 16.7 - What is the pH at the equivalence point when 25 mL...Ch. 16.7 - Prob. 16.16ECh. 16 - Write an equation for the ionization of hydrogen...Ch. 16 - Prob. 16.2QPCh. 16 - Briefly describe two methods for determining Ka...Ch. 16 - Describe how the degree of ionization of a weak...Ch. 16 - Prob. 16.5QPCh. 16 - Phosphorous acid, H2PHO3, is a diprotic acid....Ch. 16 - Prob. 16.7QPCh. 16 - Write the equation for the ionization of aniline,...Ch. 16 - Which of the following is the strongest base: NH3,...Ch. 16 - Do you expect a solution of anilinium chloride...Ch. 16 - Prob. 16.11QPCh. 16 - The pH of 0.10 M CH3NH2 (methylamine) is 11.8....Ch. 16 - Define the term buffer. Give an example.Ch. 16 - What is meant by the capacity of a buffer?...Ch. 16 - Prob. 16.15QPCh. 16 - If the pH is 8.0 at the equivalence point for the...Ch. 16 - Which of the following salts would produce the...Ch. 16 - If you mix 0.10 mol of NH3 and 0.10 mol of HCl in...Ch. 16 - Hydrogen sulfide, H2S, is a very weak diprotic...Ch. 16 - If 20.0 mL of a 0.10 M NaOH solution is added to a...Ch. 16 - Aqueous Solutions of Acids, Bases, and Salts a For...Ch. 16 - The pH of Mixtures of Acid, Base, and Salt...Ch. 16 - Which of the following beakers best represents a...Ch. 16 - You have 0.10-mol samples of three acids...Ch. 16 - Prob. 16.25QPCh. 16 - You have the following solutions, all of the same...Ch. 16 - Prob. 16.27QPCh. 16 - A chemist prepares dilute solutions of equal molar...Ch. 16 - Prob. 16.29QPCh. 16 - Prob. 16.30QPCh. 16 - You are given the following acidbase titration...Ch. 16 - The three flasks shown below depict the titration...Ch. 16 - Write chemical equations for the acid ionizations...Ch. 16 - Write chemical equations for the acid ionizations...Ch. 16 - Acrylic acid, whose formula is HC3H3O2 or...Ch. 16 - Heavy metal azides, which are salts of hydrazoic...Ch. 16 - Boric acid, B(OH)3, is used as a mild antiseptic....Ch. 16 - Formic acid, HCHO2, is used to make methyl formate...Ch. 16 - C6H4NH2COOH, para-aminobenzoic acid (PABA), is...Ch. 16 - Barbituric acid. HC4H3N2O3, is used to prepare...Ch. 16 - A solution of acetic acid, HC2H3O2, on a...Ch. 16 - A chemist wanted to determine the concentration of...Ch. 16 - Hydrofluoric acid, HF, unlike hydrochloric acid,...Ch. 16 - Chloroacetic acid, HC2H2ClO2, has a greater acid...Ch. 16 - What is the hydronium-ion concentration of a 2.00...Ch. 16 - What is the hydronium-ion concentration of a 3.00 ...Ch. 16 - Phthalic acid, H2C8H4O4, is a diprotic acid used...Ch. 16 - Carbonic acid, H2CO3, can be found in a wide...Ch. 16 - Write the chemical equation for the base...Ch. 16 - Write the chemical equation for the base...Ch. 16 - Butylamine, C4H3NH2 is a weak base. A 0.47 M...Ch. 16 - Trimethylamine, (CH3)3N, is a gas with a fishy,...Ch. 16 - What is the concentration of hydroxide ion in a...Ch. 16 - What is the concentration of hydroxide ion in a...Ch. 16 - Note whether hydrolysis occurs for each of the...Ch. 16 - Note whether hydrolysis occurs for each of the...Ch. 16 - Prob. 16.57QPCh. 16 - Prob. 16.58QPCh. 16 - For each of the following salts, indicate whether...Ch. 16 - Note whether the aqueous solution of each of the...Ch. 16 - Decide whether solutions of the following salts...Ch. 16 - Decide whether solutions of the following salts...Ch. 16 - Obtain a the Kb value for NO2; b the Ka value for...Ch. 16 - Prob. 16.64QPCh. 16 - What is the pH of a 0.025 M aqueous solution of...Ch. 16 - Calculate the OH concentration and pH of a 0.0025...Ch. 16 - Calculate the concentration of pyridine, C5H5N, in...Ch. 16 - What is the pH of a 0.30 M solution of...Ch. 16 - Calculate the degree of ionization of a 0.75 M HF...Ch. 16 - Calculate the degree of ionization of a 0.22 M...Ch. 16 - What is the pH of a solution that is 0.600 M HCHO2...Ch. 16 - What is the pH of a solution that is 0.20 M KOCN...Ch. 16 - What is the pH of a solution that is 0.10 M CH3NH2...Ch. 16 - What is the pH of a solution that is 0.15 M...Ch. 16 - A buffer is prepared by adding 39.8 mL of 0.75 M...Ch. 16 - A buffer is prepared by adding 115 mL of 0.30 M...Ch. 16 - What is the pH of a buffer solution that is 0.10 M...Ch. 16 - A buffer is prepared by mixing 525 mL of 0.50 M...Ch. 16 - What is the pH of a buffer solution that is 0.15 M...Ch. 16 - What is the pH of a buffer solution that is 0.10 M...Ch. 16 - What is the pH of a buffer solution that is 0.15 M...Ch. 16 - What is the pH of a buffer solution that is 0.15 M...Ch. 16 - How many moles of sodium acetate must be added to...Ch. 16 - How many moles of hydrofluoric acid, HF, must be...Ch. 16 - What is the pH of a solution in which 15 mL of...Ch. 16 - What is the pH of a solution in which 35 mL of...Ch. 16 - A 1.24-g sample of benzoic acid was dissolved in...Ch. 16 - A 0.400-g sample of propionic acid was dissolved...Ch. 16 - Find the pH of the solution obtained when 32 mL of...Ch. 16 - What is the pH at the equivalence point when 22 mL...Ch. 16 - A 50.0-mL sample of a 0.100 M solution of NaCN is...Ch. 16 - Sodium benzoate, NaC7H5O2, is used as a...Ch. 16 - Calculate the pH of a solution obtained by mixing...Ch. 16 - Calculate the pH of a solution obtained by mixing...Ch. 16 - Salicylic acid, C6H4OHCOOH, is used in the...Ch. 16 - Cyanoacetic acid, CH2CNCOOH, is used in the...Ch. 16 - A 0.050 M aqueous solution of sodium hydrogen...Ch. 16 - A 0.10 M aqueous solution of sodium dihydrogen...Ch. 16 - Prob. 16.99QPCh. 16 - Calculate the base-ionization constants for PO43...Ch. 16 - Calculate the pH of a 0.072 M aqueous solution of...Ch. 16 - Calculate the pH of a 0.10 M aqueous solution of...Ch. 16 - An artificial fruit beverage contains 11.0 g of...Ch. 16 - A buffer is made by dissolving 12.5 g of sodium...Ch. 16 - Blood contains several acid base systems that tend...Ch. 16 - Codeine, C23H21NO3, is an alkaloid (Kb = 6 2 109)...Ch. 16 - Calculate the pH of a solution obtained by mixing...Ch. 16 - Calculate the pH of a solution made up from 2.0 g...Ch. 16 - Find the pH of the solution obtained when 25 mL of...Ch. 16 - What is the pH of the solution obtained by...Ch. 16 - Ionization of the first proton from H2SO4 is...Ch. 16 - Ionization of the first proton from H2SeO4 is...Ch. 16 - Methylammonium chloride is a salt of methylamine,...Ch. 16 - Sodium benzoate is a salt of benzoic acid,...Ch. 16 - Each of the following statements concerns a 0.010...Ch. 16 - Each of the following statements concerns a 0.10 M...Ch. 16 - A 0.288-g sample of an unknown monoprotic organic...Ch. 16 - A 0.239-g sample of unknown organic base is...Ch. 16 - a Draw a pH titration curve that represents the...Ch. 16 - a Draw a pH titration curve that represents the...Ch. 16 - The equilibrium equations and Ka values for three...Ch. 16 - Prob. 16.122QPCh. 16 - A 25.0-mL sample of hydroxylamine is titrated to...Ch. 16 - A 25.00-mL sample contains 0.562 g of NaHCO3. This...Ch. 16 - A solution made up of 1.0 M NH3 and 0.50 M...Ch. 16 - A solution is prepared from 0.150 mol of formic...Ch. 16 - An important component of blood is the buffer...Ch. 16 - An important component of blood is the buffer...Ch. 16 - Tartaric acid is a weak diprotic fruit acid with...Ch. 16 - Malic acid is a weak diprotic organic acid with...Ch. 16 - A quantity of 0.25 M sodium hydroxide is added to...Ch. 16 - A quantity of 0.15 M hydrochloric acid is added to...Ch. 16 - Prob. 16.133QPCh. 16 - Prob. 16.134QPCh. 16 - A 30.0-mL sample of 0.05 M HClO is titrated by a...Ch. 16 - Prob. 16.136QPCh. 16 - Prob. 16.137QPCh. 16 - Calculate the pH of a solution made by mixing 0.62...Ch. 16 - Cyanic acid, HOCN, is a weak acid with a Ka value...Ch. 16 - The Kb for NH3 is 1.8 105 at 25C. Calculate the...Ch. 16 - Ka for formic acid is 1.7 104 at 25C. A buffer is...Ch. 16 - K4 for acetic acid is 1.7 105 at 25C. A buffer...Ch. 16 - Prob. 16.143QPCh. 16 - Prob. 16.144QPCh. 16 - Prob. 16.145QPCh. 16 - Two samples of 1.00 M HCl of equivalent volumes...Ch. 16 - Prob. 16.147QPCh. 16 - Prob. 16.148QPCh. 16 - A solution of weak base is titrated to the...Ch. 16 - A buffer solution is prepared by mixing equal...Ch. 16 - The pH of a white vinegar solution is 2.45. This...Ch. 16 - The pH of a household cleaning solution is 11.50....Ch. 16 - What is the freezing point of 0.92 M aqueous...Ch. 16 - Prob. 16.154QPCh. 16 - A chemist needs a buffer with pH 4.35. How many...Ch. 16 - A chemist needs a buffer with pH 3.50. How many...Ch. 16 - Weak base B has a pKb of 6.78 and weak acid HA has...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Two samples of 1.00 M HCl of equivalent volumes are prepared. One sample is titrated to the equivalence point with a 1.00 M solution of sodium hydroxide, while the other sample is titrated to the equivalence point with a 1.00 M solution of calcium hydroxide. a Compare the volumes of sodium hydroxide and calcium hydroxide required to reach the equivalence point for each titration. b Determine the pH of each solution halfway to the equivalence point. c Determine the pH of each solution at the equivalence point.arrow_forwardMethyl orange, HMO, is a common acid-base indicator. In solution it ionizes according to the equation: HMOaqH+aq+MO-aqredyellow If methyl orange is added to distilled water, the solution turns yellow. If 1 drop or two of 6 M HCl is added to the yellow solution, it turns red. If to that solution one adds a few drops of 6 M NaOH, the color reverts to yellow. a. Why does adding 6 M HCl to the yellow solution of methyl orange tend to cause the color to change to red? Note that in solution HCl exists as H+ and Cl- ions. b. Why does adding 6 M NaOH to the red solution tend to make it turn back to yellow? Note that in solution NaOH exists as Na+ and OH- ions. How does increasing OH- shift Reaction 3 in the discussion section? How would the resulting change in H+ affect the dissociation reaction of HMO?arrow_forwardA solution of weak base is titrated to the equivalence point with a strong acid. Which one of the following statements is most likely to be correct? a The pH of the solution at the equivalence point is 7.0. b The pH of the solution is greater than 13.0. c The pH of the solution is less than 2.0. d The pH of the solution is between 2.0 and 7.0. e The pH of the solution is between 7.0 and 13.0. The reason that best supports my choosing the answer above is a Whenever a solution is titrated with a strong acid, the solution will be very acidic. b Because the solution contains a weak base and the acid (titrant) is used up at the equivalence point, the solution will be basic. c Because the solution contains the conjugate acid of the weak base at the equivalence point, the solution will be acidic.arrow_forward
- Enough water is added to the buffer in Question 30 to make the total volume 5.00 L. (a) Calculate the pH of the buffer. (b) Calculate the pH of the buffer after adding 0.0250 mol of HCl to 0.376 L of the buffer. (c) Calculate the pH of the buffer after adding 0.0250 mol of KOH to 0.376 L of the buffer. (d) Compare your answers to Question 30 (a-c) with your answers to (a-c) of this problem. (e) Comment on the effect of dilution on the pH of a buffer and on its buffer capacity. Âarrow_forwardUse the same symbols as in Question 61 ( = anion, =OH) for the box below. (a) Fill in a similar box (representing one liter of the same solution) after 2 mol of H+ (2) have been added. Indicate whether the resulting solution is an acid, base, or buffer. (b) Follow the directions of part (a) for the resulting solution after 2 mol of OH- (2 ) have been added. (c) Follow the directions of part (a) for the resulting solution after 5 mol of OH- (5 ) have been added. (Hint: Write the equation for the reaction before you draw the results.)arrow_forwardUsing the diagrams shown in Problem 10-117, which of the solutions would have the greatest buffer capacity, that is, greatest protection against pH change, when the following occurs? a. A strong acid is added to the solution. b. A strong base is added to the solution.arrow_forward
- Consider all acid-base indicators discussed in this chapter. Which of these indicators would be suitable for the titration of each of these? (a) NaOH with HClO4 (b) acetic acid with KOH (c) NH3 solution with HBr (d) KOH with HNO3 Explain your choices.arrow_forward. Write the formulas for three combinations of weak acid and salt that would act as buffered solutions. For each of your combinations, write chemical equations showing how the components of the buffered solution would consume added acid and base.arrow_forwardA weak acid, HA, is dissolved in water. Which one of the following beakers represents the resulting solution? (Water molecules have been omitted for clarity.)arrow_forward
- Four different substances of the generalized formula HA were dissolved in water, with the results shown in the diagrams. Which of the diagrams represents the substance that is the strongest electrolyte?arrow_forwardA quantity of 0.15 M hydrochloric acid is added to a solution containing 0.10 mol of sodium acetate. Some of the sodium acetate is converted to acetic acid, resulting in a final volume of 650 mL of solution. The pH of the final solution is 4.56. a What is the molar concentration of the acetic acid? b How many milliliters of hydrochloric acid were added to the original solution? c What was the original concentration of the sodium acetate?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning


Chemical Principles in the Laboratory
Chemistry
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Brooks Cole

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Acid-Base Equilibrium; Author: Bozeman Science;https://www.youtube.com/watch?v=l5fk7HPmo5g;License: Standard YouTube License, CC-BY
Introduction to Titrimetric analysis; Author: Vidya-mitra;https://www.youtube.com/watch?v=uykGVfn9q24;License: Standard Youtube License