
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition (8th Edition)
8th Edition
ISBN: 9780134218328
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 16, Problem 16.43AP
Interpretation Introduction
Interpretation:
It should be explained that why the given ammonium salt does not reacts with acid and base.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
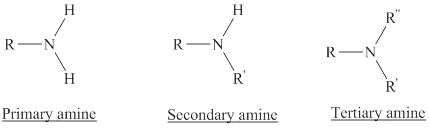
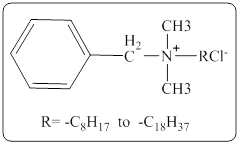
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
One commonly encountered quaternary ammonium salt has the following structure,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Tetrabromofluorescein is a purple dye often used in lipsticks. if the dye is purple, what color does it absorb?
Explain the difference in the melting points of the linoleic acid and linolenic acid:
The shape of hair is determined in part by the pattern of disulfide bonds in keratin, its major protein. How can curls be induced? Explain chemically the basic steps (type of chemical and order) of what a hairstylist would use to treat the hair.
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition (8th Edition)
Ch. 16.2 - Identify the following compounds as primary,...Ch. 16.2 - Prob. 16.2PCh. 16.2 - Prob. 16.3PCh. 16.2 - Prob. 16.4PCh. 16.2 - Prob. 16.5KCPCh. 16.2 - Prob. 16.6KCPCh. 16.3 - Arrange the following compounds in order of...Ch. 16.3 - Draw the structures of (a) ethylamine and (b)...Ch. 16.4 - Provide compounds that fit the following...Ch. 16.4 - Prob. 16.10P
Ch. 16.4 - Prob. 16.11PCh. 16.5 - Write an equation for the acid-base equilibrium...Ch. 16.5 - Prob. 16.13PCh. 16.5 - Prob. 16.14PCh. 16.5 - Prob. 16.15PCh. 16.5 - Prob. 16.16PCh. 16.6 - Prob. 16.17PCh. 16.6 - Prob. 16.18PCh. 16.6 - Prob. 16.19PCh. 16.6 - Prob. 16.20PCh. 16.6 - Prob. 16.21PCh. 16.6 - Prob. 16.22PCh. 16.7 - Prob. 16.1CIAPCh. 16.7 - Prob. 16.2CIAPCh. 16.7 - Prob. 16.3CIAPCh. 16 - (a) For the compound above, identify each nitrogen...Ch. 16 - The structure of the amino acid lysine (in its...Ch. 16 - Prob. 16.25UKCCh. 16 - Prob. 16.26UKCCh. 16 - Prob. 16.27UKCCh. 16 - Complete the following equations: (a) (b)...Ch. 16 - Prob. 16.29APCh. 16 - Draw the structures corresponding to the following...Ch. 16 - Name the following amines, and classify them as...Ch. 16 - Name the following amines, and identify them as...Ch. 16 - Prob. 16.33APCh. 16 - Which is a stronger base, diethyl ether or...Ch. 16 - Prob. 16.35APCh. 16 - Prob. 16.36APCh. 16 - The compound lidocaine is used medically as a...Ch. 16 - Prob. 16.38APCh. 16 - Draw the structures of the ammonium ions formed...Ch. 16 - Prob. 16.40APCh. 16 - Prob. 16.41APCh. 16 - Prob. 16.42APCh. 16 - Prob. 16.43APCh. 16 - Prob. 16.44APCh. 16 - Prob. 16.45CPCh. 16 - Prob. 16.46CPCh. 16 - Prob. 16.47CPCh. 16 - Prob. 16.48CPCh. 16 - How do amines differ from analogous alcohols in...Ch. 16 - Name at least two undesirable characteristics are...Ch. 16 - Prob. 16.52CPCh. 16 - Complete the following equations (Hint: Answers...Ch. 16 - Prob. 16.54CPCh. 16 - Prob. 16.55CPCh. 16 - Why is cyclohexylamine not considered to be a...Ch. 16 - Prob. 16.57CPCh. 16 - Prob. 16.58GPCh. 16 - 1-Propylamine, 1-propanol, acetic acid, and butane...Ch. 16 - Prob. 16.60GPCh. 16 - Lemon juice, which contains citric acid, is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- For each of the following chemicals, name the general class they belong to, discuss their solubility in water and explain why they are/are not soluble in water (note the numbers are all subscripts): a) CH3(CH2)26COOH b) KCl c) CH3OHarrow_forwardAn amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper chromatography.The solvent is less polar than water. Which of these amino acids will have the highest Rf value and which the lowest? Explain.arrow_forwardAn amino acid mixture of phenylalanine, glycine and glutamic acid is to be separated by paper chromatography. The solvent is less polar than water. Which of these amino acids will have the highest Rf value and which the lowest? Explain.arrow_forward
- "the same tripeptide" pertains to the tripeptide, ILYarrow_forwardWhich of the following is removed when tea is brewed in hot water? A. Cellulose B. Tannins C. Glucose D. Caffeine E. Sodium bicarbonatearrow_forwardHow many mL of methadone is needed for a patient who was prescribed 0.015 g of methadone? The vial reads 6 milligrams of methadone per 1 mL of solution.arrow_forward
- If water were not polar, would it dissolve sodium chloridearrow_forwardWhich of the following ingredients in Ding Dong Cakes are added for the purpose of maintaining pH until you consume them: a. sodium chloride b. sodium citrate/citric acid c. sodium malate/malic acid d. monosodium phosphate/disodium phosphate e. lecithinarrow_forwardWhich acid in each pair has the higher melting point and explain why Arachidonic acid or Archidic acid.arrow_forward
- Why Cyanide Is So Deadly?arrow_forwardWhat is the isoelectric point of casein? Please choose one correct answer only. A. pH 6.6 B. pH 5.6 C. pH 4.6 D. pH 3.6 E. None of the given optionsarrow_forwardIf the dehydration reaction of an alcohol is successful, what changes would be seen in the IR spectrum for the product compared to the starting materialarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
- Essentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage



Essentials of Pharmacology for Health Professions
Nursing
ISBN:9781305441620
Author:WOODROW
Publisher:Cengage

GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license