
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
9th Edition
ISBN: 9781337580632
Author: Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.9E
Give each of the following
a. 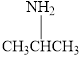
b. 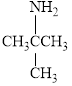
c. 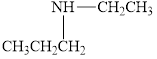
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Use the structure of aniline provided and draw the condensedformula for each of the following amines.a. N-Methylaniline c. N-Ethylanilineb. N,N-Dimethylaniline d. N-Isopropylaniline
Cyclic amides are called which of the following?
a. lactams b. lactanes c. lactones d. lactines
Explain why many amines with useful medicinal properties are sold as their ammonium salts
Chapter 16 Solutions
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
Ch. 16 - Prob. 16.1ECh. 16 - Prob. 16.2ECh. 16 - Prob. 16.3ECh. 16 - Prob. 16.4ECh. 16 - Prob. 16.5ECh. 16 - Prob. 16.6ECh. 16 - Prob. 16.7ECh. 16 - Prob. 16.8ECh. 16 - Give each of the following amines an IUPAC name:...Ch. 16 - Give each of the following amines an IUPAC name....
Ch. 16 - Prob. 16.11ECh. 16 - Prob. 16.12ECh. 16 - Prob. 16.13ECh. 16 - Prob. 16.14ECh. 16 - Prob. 16.15ECh. 16 - Prob. 16.16ECh. 16 - Prob. 16.17ECh. 16 - Prob. 16.18ECh. 16 - Prob. 16.19ECh. 16 - Draw diagrams similar to Figure 16.1 to illustrate...Ch. 16 - Prob. 16.21ECh. 16 - Prob. 16.22ECh. 16 - Prob. 16.23ECh. 16 - Prob. 16.24ECh. 16 - Prob. 16.25ECh. 16 - Prob. 16.26ECh. 16 - Prob. 16.27ECh. 16 - Prob. 16.28ECh. 16 - Prob. 16.29ECh. 16 - Prob. 16.30ECh. 16 - Prob. 16.31ECh. 16 - Prob. 16.32ECh. 16 - Prob. 16.33ECh. 16 - Describe the general structure of a neuron.Ch. 16 - Name the two amino acids that are starting...Ch. 16 - Prob. 16.36ECh. 16 - Prob. 16.37ECh. 16 - Prob. 16.38ECh. 16 - Prob. 16.39ECh. 16 - Prob. 16.40ECh. 16 - Prob. 16.41ECh. 16 - Prob. 16.42ECh. 16 - Why are alkaloids weakly basic?Ch. 16 - Prob. 16.44ECh. 16 - Prob. 16.45ECh. 16 - Prob. 16.46ECh. 16 - Prob. 16.47ECh. 16 - Prob. 16.48ECh. 16 - Prob. 16.49ECh. 16 - Prob. 16.50ECh. 16 - Prob. 16.51ECh. 16 - Complete the following reactions: a. b.Ch. 16 - Complete the following reactions: a. b.Ch. 16 - Prob. 16.54ECh. 16 - What are the products of the acid hydrolysis of...Ch. 16 - Prob. 16.56ECh. 16 - Prob. 16.57ECh. 16 - Prob. 16.58ECh. 16 - Prob. 16.59ECh. 16 - Prob. 16.60ECh. 16 - Prob. 16.61ECh. 16 - Prob. 16.62ECh. 16 - Prob. 16.63ECh. 16 - Prob. 16.64ECh. 16 - Prob. 16.65ECh. 16 - Prob. 16.66ECh. 16 - Prob. 16.67ECh. 16 - Prob. 16.68ECh. 16 - Prob. 16.69ECh. 16 - Prob. 16.70ECh. 16 - Prob. 16.71ECh. 16 - The stimulant in coffee is: a. tannic acid b....Ch. 16 - What are the most likely products of a reaction...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 12:58 Question 15.a of 25 Classify the following amine. A) primary amine B) secondary amine C) tertiary amine D) quaternary ammonium salt CH3 -Z N Submit / Periodic Table Harrow_forwardDraw a structural formula for each amine and amine derivative. Q.)1-Phenyl-2-propanamine (amphetamine)arrow_forwardDraw a structural formula for each amine and amine derivative. Q.) N,N-Dimethylanilinearrow_forward
- The hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forwardDraw a structural formula for each amine and amine derivative. Q.) Lithium diisopropylamide (LDA)arrow_forwardFive examples each of organic acids, esters, amines, amides and alkaloids with their IUPAC name, structure, and uses.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY