
World of Chemistry
7th Edition
ISBN: 9780618562763
Author: Steven S. Zumdahl
Publisher: Houghton Mifflin College Div
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16.1, Problem 5RQ
Interpretation Introduction
Interpretation: To label which species acts as Brønsted-Lowry acid-base pair.
Concept introduction: A Brønsted-Lowry acid is a hydrogen ion (
A species that can as act both as an acid and as a base according to Brønsted-Lowry Theory is said to be amphoteric. Since water can act both as an acid and as a base, water is amphoteric.
Expert Solution & Answer
Answer to Problem 5RQ
Explanation of Solution
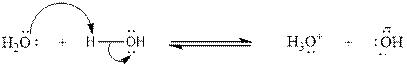
As, we can see from the reaction mechanism that one molecule of
Conclusion
Chapter 16 Solutions
World of Chemistry
Ch. 16.1 - Prob. 1RQCh. 16.1 - Prob. 2RQCh. 16.1 - Prob. 3RQCh. 16.1 - Prob. 4RQCh. 16.1 - Prob. 5RQCh. 16.1 - Prob. 6RQCh. 16.2 - Prob. 1RQCh. 16.2 - Prob. 2RQCh. 16.2 - Prob. 3RQCh. 16.2 - Prob. 4RQ
Ch. 16.2 - Prob. 5RQCh. 16.2 - Prob. 6RQCh. 16.2 - Prob. 7RQCh. 16.3 - Prob. 1RQCh. 16.3 - Prob. 2RQCh. 16.3 - Prob. 3RQCh. 16.3 - Prob. 4RQCh. 16.3 - Prob. 5RQCh. 16.3 - Prob. 6RQCh. 16 - Prob. 1ACh. 16 - Prob. 2ACh. 16 - Prob. 3ACh. 16 - Prob. 4ACh. 16 - Prob. 5ACh. 16 - Prob. 6ACh. 16 - Prob. 7ACh. 16 - Prob. 8ACh. 16 - Prob. 9ACh. 16 - Prob. 10ACh. 16 - Prob. 11ACh. 16 - Prob. 12ACh. 16 - Prob. 13ACh. 16 - Prob. 14ACh. 16 - Prob. 15ACh. 16 - Prob. 16ACh. 16 - Prob. 17ACh. 16 - Prob. 18ACh. 16 - Prob. 19ACh. 16 - Prob. 20ACh. 16 - Prob. 21ACh. 16 - Prob. 22ACh. 16 - Prob. 23ACh. 16 - Prob. 24ACh. 16 - Prob. 25ACh. 16 - Prob. 26ACh. 16 - Prob. 27ACh. 16 - Prob. 28ACh. 16 - Prob. 29ACh. 16 - Prob. 30ACh. 16 - Prob. 31ACh. 16 - Prob. 32ACh. 16 - Prob. 33ACh. 16 - Prob. 34ACh. 16 - Prob. 35ACh. 16 - Prob. 36ACh. 16 - Prob. 37ACh. 16 - Prob. 38ACh. 16 - Prob. 39ACh. 16 - Prob. 40ACh. 16 - Prob. 41ACh. 16 - Prob. 42ACh. 16 - Prob. 43ACh. 16 - Prob. 44ACh. 16 - Prob. 45ACh. 16 - Prob. 46ACh. 16 - Prob. 47ACh. 16 - Prob. 48ACh. 16 - Prob. 49ACh. 16 - Prob. 50ACh. 16 - Prob. 51ACh. 16 - Prob. 52ACh. 16 - Prob. 53ACh. 16 - Prob. 54ACh. 16 - Prob. 55ACh. 16 - Prob. 56ACh. 16 - Prob. 57ACh. 16 - Prob. 58ACh. 16 - Prob. 59ACh. 16 - Prob. 60ACh. 16 - Prob. 61ACh. 16 - Prob. 62ACh. 16 - Prob. 1STPCh. 16 - Prob. 2STPCh. 16 - Prob. 3STPCh. 16 - Prob. 4STPCh. 16 - Prob. 5STPCh. 16 - Prob. 6STPCh. 16 - Prob. 7STPCh. 16 - Prob. 8STPCh. 16 - Prob. 9STPCh. 16 - Prob. 10STPCh. 16 - Prob. 11STP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY