
(a)
Interpretation: The given Fischer esterification reaction in which the alcohol is present in excess should be completed.
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,
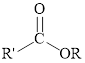
Fischer esterification can be represented as follows,
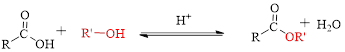
(b)
Interpretation: The given Fischer esterification reaction in which the alcohol is present in excess should be completed.
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,

Fischer esterification can be represented as follows,
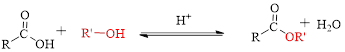
(c)
Interpretation: The given Fischer esterification reaction in which the alcohol is present in excess should be completed.
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,

Fischer esterification can be represented as follows,
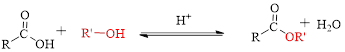
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- Would you expect hemiacetals to be stable in basic solutions? Explain your answer.arrow_forwardProvide a short paragraph that relates the most essential elements of theory behind the fischer Esterification reaction.arrow_forwardSynthesising menthone is an exothermic process that includes a reflux procedure. Mark the correct statements. * A- The reflux process enables us to transfers energy to the reaction mixture for an extended period of time without loss of solvent or reagents. B- The reflux process increases the rate of the oxidation of menthol. C- The reflux process increases the yield of the oxidation of menthol. D- A water bath can be used in the preparation of menthone because the boiling point of the reaction mixture is below 100°C due to the inclusion of acetone in this mixture.arrow_forward
- A chemist is performing a Fischer Esterification but can't find the sulfuric acid. This reagent is important because itarrow_forwardDraw the molecular structure of p-nitrophenol in an acidic solution and in an alkaline solution.arrow_forwardWhat is the difference between propanol-water IMFs compared to water-water and propanol-propanol IMFs?arrow_forward
- a)What happens when isoamyl alcohol is allowed to heated with Acetic Acid in presence of concentrated sulphuric acid? What is role of concentrated sulphuric acid?arrow_forwardIn what proportion should alcohols of 80% and 35% strengths be mixed to make60% alcohol?arrow_forwardDraw a structural formula for this cyclic hemiacetal.arrow_forward
- According to Le Chatelier's Principle, acetal product formation tends to INCREASE (rather than decrease) when more alcohol is added to the reaction mixture. True or False?arrow_forwardExplain differences in the UV vis spectra between cinnamaldehyde, cinnamic acid, cinnamyl alcohol, 3-phenylpropanalarrow_forwardThe cyclic hemiacetal is more stable than the open-chain form, so very little of the open-chain form is present atequilibrium. Will an aqueous solution of glucose reduce Tollens reagent and give a positive Tollens test? Explain.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

