
(a)
Interpretation: The equation for the reaction of acid in Example 17.3 with ammonia has to be written and the name of the carboxylic acid salt formed should be named.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
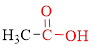
Nomenclature of carboxylic acid:
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the
alkane name with –oic acid.
Carboxylic acid reacts with ammonia gives neutral carboxylic salt.
(b)
Interpretation: The equation for the reaction of acid in Example 17.3 with ammonia has to be written and the name of the carboxylic acid salt formed should be named.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,

Nomenclature of carboxylic acid:
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the alkane name with –oic acid.
Carboxylic acid reacts with ammonia gives neutral carboxylic salt.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry
- Write an equation for the reaction of chloroacetic acid (Ka=1.5103) with trimethylamine (Kb=5.9105) . Calculate the equilibrium constant for the reaction. If 0.10 M solutions of these two species are mixed, what will be their concentrations at equilibrium?arrow_forwardWrite the structural formulas of trichloroacetic acid and acetic acid and explain why trichloroacetic acid is the much stronger acid of the two.arrow_forwardAcetic acid, salicylic acid, benzoic acid all of them undergo esterification test separately with n-propyl alcohol and concentrated sulfuric acid. Which among the three is the fastest in showing the formation of pleasant odor. Arrange them from fastest to slowest. Explain and support your answerarrow_forward
- Upon completion of a chemical reaction, you find you have a mixture of benzoic acid and ethyl benzoate. Propose a procedure to separate the ethyl benzoate from the mixture. You should look up the structures of benzoic acid and ethyl benzoate.arrow_forwardIn the Separating Acids and Neutral Compounds by Solvent Extraction experiment, why was sodium sulfate added to the tert-butyl methyl ether layer ? Group of answer choices to acidify the compounds to deprotonate the compounds to precipitate the compounds to remove traces of waterarrow_forwardwrite the reactions in which one of the following compounds acts on the ammonia solution of silver oxide. a) propene b) bytine-2 c) cyclopropane d) pentin-1arrow_forward
- What is the chemical equation of the reaction of Ethanol with Acetic acid in the presence of sulfuric acidarrow_forwardConcentrated sulfuric acid is used as a catalyst for the Fischer esterification. Why is sulfuric acid needed if another acid, acidic acid, is already present?arrow_forwardWhich of the following statements is true? a) Replacing bromine with chlorine does not affect the rate of elimination b) The speed of the reaction depends on the concentration of ethanol c) The reaction is completely irreversible d) It is an equilibrium reaction that can also occur in the opposite directionarrow_forward
- Describe the trends in the physical properties of carboxylic acids, and contrast theirphysical properties with those of their salts.arrow_forwardThe formate and methoxide ions are both formed by removing a hydrogen atom from a corresponding neutral compound. Given that each process involves the breaking of an O-H bond, explain why the formate ion is so much more stable than the methoxide ion.arrow_forwardWhy is benzoic acid insoluble in sulfuric acid? Explain the conceptarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


