
(a)
Interpretation: Mechanism for the given type of decarboxylation reaction has to be proposed and it should be compared with the mechanism of decarboxylation of
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
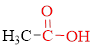
If carboxylic acid is heated to a very high temperature then of carbon dioxide will be eliminated from it this reaction is known as decarboxylation. Simple carboxylic acids do not decarboxylate readily.
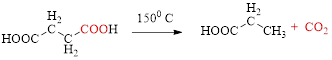
Decarboxylation of
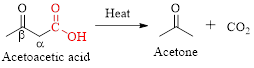
The mechanism of thermal decarboxylation involves two processes,
- (i) Redistribution of electrons in a cyclic transition state.
- (ii) Cyclic transition state possesses keto-enol tautomerism.
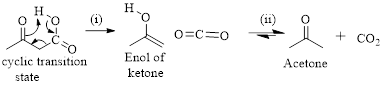
(b)
Interpretation: Mechanism for the given type of decarboxylation reaction has to be proposed and it should be compared with the mechanism of decarboxylation of
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,

If carboxylic acid is heated to a very high temperature then of carbon dioxide will be eliminated from it this reaction is known as decarboxylation. Simple carboxylic acids do not decarboxylate readily.
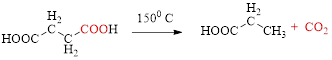
Decarboxylation of
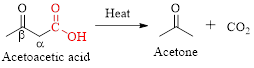
Simply the mechanism of thermal decarboxylation involves two processes,
- (i) Redistribution of electrons in a cyclic transition state.
- (ii) Cyclic transition state possesses keto-enol tautomerism.
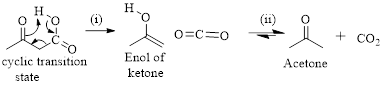
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- -Hydroxyketones and -hydroxyaldehydes are also oxidized by treatment with periodic acid. It is not the -hydroxyketone or aldehyde, however, that undergoes reaction with periodic acid, but the hydrate formed by addition of water to the carbonyl group of the -hydroxyketone or aldehyde. Write a mechanism for the oxidation of this -hydroxyaldehyde by HIO4.arrow_forwardThe base-promoted rearrangement of an -haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. It is proposed that NaOH first converts the a-haloketone to the substituted cyclopropanone shown in brackets and then to the sodium salt of cyclopentanecarboxylic acid. (a) Propose a mechanism for base-promoted conversion of 2-chlorocyclohexanone to the proposed intermediate. (b) Propose a mechanism for base-promoted conversion of the proposed intermediate to sodium cyclopentanecarboxylate.arrow_forwardA chemist in need of 2,2-dimethylpentanoic acid decided to synthesize some by reaction of 2-chloro-2-methylpentane with NaCN, followed by hydrolysis of the product. After the reaction sequence was carried out, however, none of the desired product could be found. What do you suppose went wrong?arrow_forward
- The following bicyclic ketone has two -carbons and three -hydrogens. When this molecule is treated with D2O in the presence of an acid catalyst, only two of the three -hydrogens exchange with deuterium. The -hydrogen at the bridgehead does not exchange. How do you account for the fact that two -hydrogens do exchange but the third does not? You will find it helpful to build models of the enols by which exchange of -hydrogens occurs.arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardHow does hcl react with this compound via acid-catalyzed electophilic aromatic substitution-alkylation?arrow_forward
- Propose a mechanism for the bromination of ethoxybenzene to give o- and p-bromoethoxybenzenearrow_forwarda) Give the appropriate base for the above reaction and draw the resonance structures of the enolate ions derived from compound A. Then write the mechanism for the formation of the Dieckmann cyclized product B. b) If compound B above is reacted with NaBH 4 , draw the structure of the reduced product. Give a reason for your choice of product. c) Being a β-ketoester, B could undergo a 3-step synthesis involving alkylation, hydrolysis and decarboxylation reactions to yield the cyclopentanone, C. Write the outline synthesis for each step which include the appropriate regents and correct intermediate.arrow_forwardIf the following compound were treated with excess NaOH followed by an excess of D2O,what would be the structure of the new product formed?arrow_forward
- Propose a plausible arrow pushing mechanism for the following transformation. Hint: hydrolysis of acetal followed by ester formation.arrow_forwardWhether the ortho-carboxy substituent acts as an intramolecular general-base catalyst or as an intramolecular nucleophilic catalyst can be determined by carrying out the hydrolysis of aspirin with 18O-labeled water and determining whether 18O is incorporated into ortho-carboxy-substituted phenol. Explain the results that would be obtained with the two types of catalysis.arrow_forwardTo prove that the reaction of an ester with hydroxide ion is not an SN2 reaction but, instead, forms a tetrahedral intermediate that subsequently collapses, the products obtained after allowing hydroxide ion to react with an ester containing an isotopically labeled carbonyl oxygen (18O) were isolated. One of the products was isotopically labeled hydroxide ion. Explain how obtaining this product proves that a tetrahedral intermediate was formed.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

