
Concept explainers
(a)
Interpretation:
Show how to prepare pentanoic acid from 1-Pentanol.
Concept introduction:
Carboxylic acid can be prepared from various ways; oxidation of
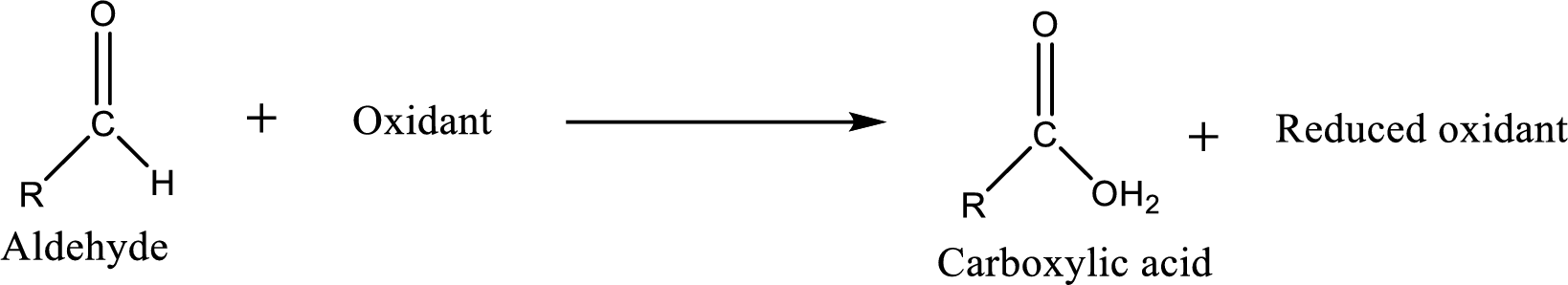
Carboxylic acid on further oxidation removes the carboxyl carbon as carbon dioxide.
Depending on the reaction conditions, the oxidation state of the remaining organic structure may be higher, lower or unchanged.
Carboxylic acid can be prepared from primary alcohol by oxidation using strong oxidizing agents like chromic acid,
(b)
Interpretation:
Show how to prepare pentanoic acid from Pentanal.
Concept introduction:
Carboxylic acid can be prepared from various ways; oxidation of aldehyde is one of the important methods to prepare carboxylic acid.
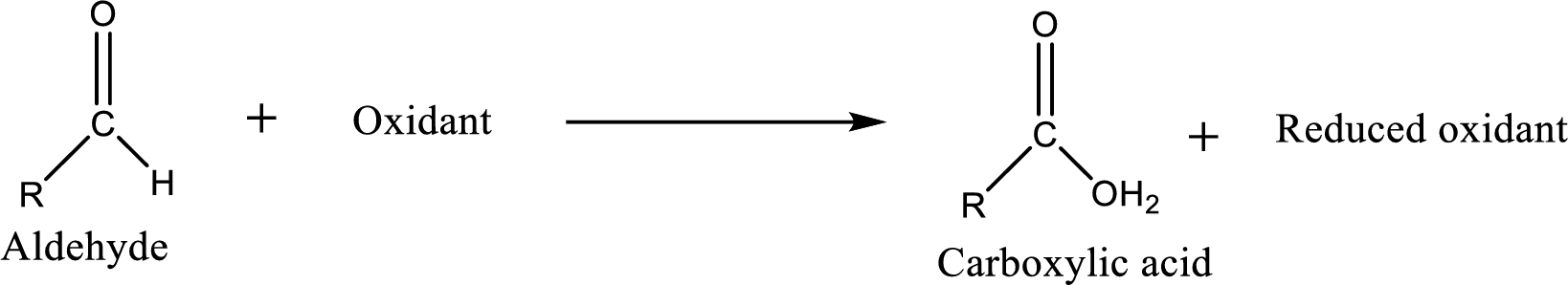
Aldehyde and
In aldehyde
In ketone
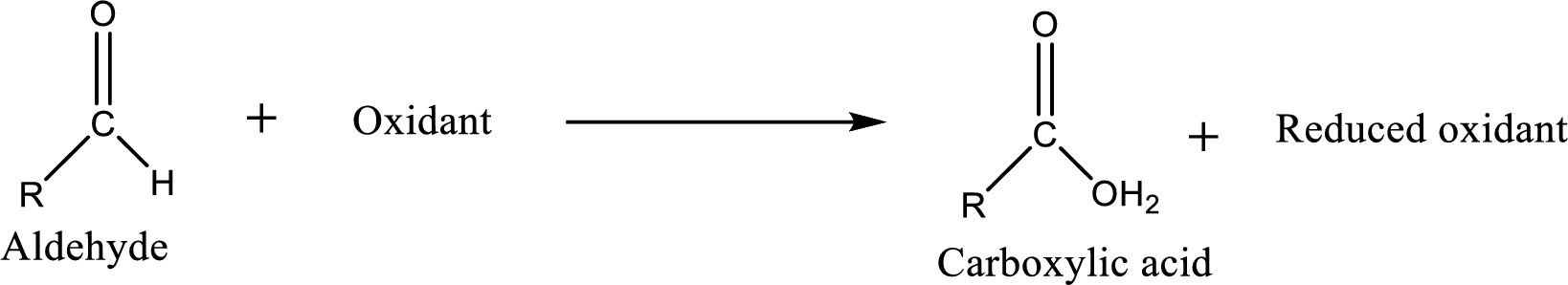
Aldehyde readily undergoes oxidation to carboxylic acids.
Tollens’ reagent is an ammoniac silver nitrate solution which can be used to detect the presence of aldehyde in an unknown compound.
As the oxidation of the aldehyde proceeds by Tollens’ reagent, silver metal is deposited on the walls of the reaction flask as a shiny mirror.
The reaction can be represented as follows,
(c)
Interpretation:
Show how to prepare pentanoic acid from 1-Pentene.
Concept introduction:
Carboxylic acid can be prepared from various ways; oxidation of aldehyde is one of the important methods to prepare carboxylic acid.
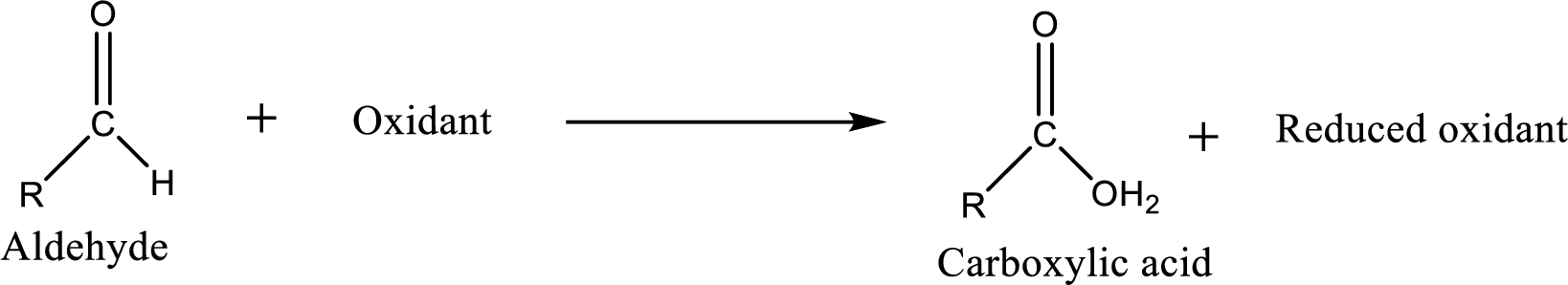
Aldehyde and ketones are one such an important group in the organic compounds. Both of these compounds contain carbonyl group
In aldehyde
In ketone
Carboxylic acid can be prepared from primary alcohol by oxidation using strong oxidizing agents like chromic acid,
Alkenes on acid catalyzed hydration will give alcohol.
(d)
Interpretation:
Show how to prepare pentanoic acid from 1-Butanol.
Concept introduction:
Alkyl or aryl magnesium halides (RMgX) are known as Grignard reagent. The Grignard reaction is an organometallic
Synthesis of Grignard reagent is shown below,
The
Addition of a Grignard reagent to carbon dioxide followed by protonation will produce carboxylic acid.
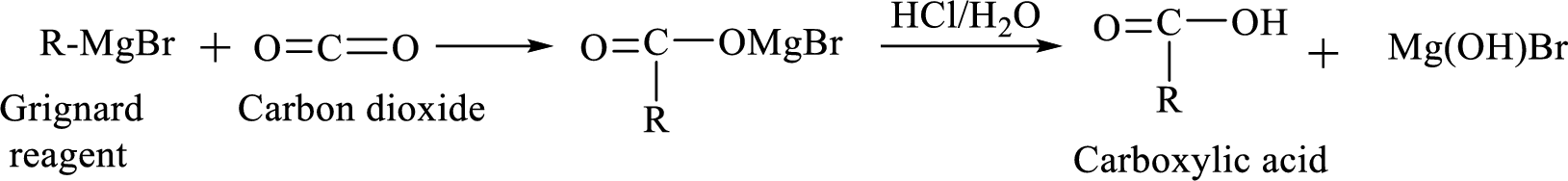
(e)
Interpretation:
Show how to prepare pentanoic acid from 1-Bromopropane.
Concept introduction:
Alkyl or aryl magnesium halides (RMgX) are known as Grignard reagent. The Grignard reaction is an organometallic chemical reaction in which the Grignard reagent act as nucleophile and attack electrophilic carbon atom that are present within polar bonds to yield a carbon-carbon bond.
Synthesis of Grignard reagent is shown below,
The alkyl halide can be prepared from alcohol through different methods, preparing alkyl halide using halogens is one of the important methods and it is shown below,
Addition of a Grignard reagent to carbon dioxide followed by protonation will produce carboxylic acid.
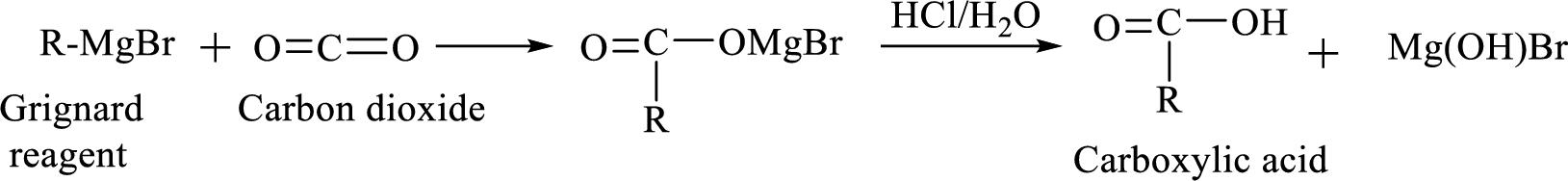
(f)
Interpretation:
Show how to prepare pentanoic acid from 1-Hexene.
Concept introduction:
Carboxylic acid can be prepared from various ways; oxidation of aldehyde is one of the important methods to prepare carboxylic acid.
Aldehyde and ketones are one such an important group in the organic compounds. Both of these compounds contain carbonyl group
In aldehyde
In ketone
The reaction of
Trending nowThis is a popular solution!
Learn your wayIncludes step-by-step video

Chapter 17 Solutions
Organic Chemistry
- (a) Draw the structures of the following compounds :(i) 4-Chloropentan-2-one (ii) p-Nitropropiophenone(b) Give tests to distinguish between the following pairs of compounds :(i) Ethanal and Propanal (ii) Phenol and Benzoic acid(iii) Benzaldehyde and Acetophenonearrow_forwardFollowing is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardThe following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forward
- Starting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare the compound Q.)1,6-Hexanediolarrow_forwardWrite reactions of ethyl chloride with the following reagents: a. KOH, aqueous solution b. NaCNarrow_forwardBenzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.arrow_forward
- A. In the synthesis of 1-bromobutane, what is the inorganic by-product left in the reaction flask following the distillation? Why was the bromoalkane the bottom layer in the separatory funnel? B. Predict the product when 1-methylcyclohexanol reacts with H2SO4 and KBr. Show the mechanism.arrow_forwardWrite equations for the reaction of 1-butanol with reagent. Where you predict no reaction, write NR. Q) K2Cr2O7, H2SO4, H2O, heatarrow_forwardMention and Write the following reaction mechanisms: a) thermal dehydration of 4-t-butylcyclohexanol with phosphoric acid as catalyst; b) addition of molecular bromine to the C= C double bond of 4-t- butylcyclohexene;arrow_forward
- Draw a structural formula for the product formed by treating butanal with reagent. Q.) LiAlH4 followed by H2Oarrow_forward2-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O toform compound W. Compound X and compound Y are produced when 2-bromo-2-methylbutane undergoes elimination reaction with alcoholic ofsodium hydroxide, NaOH. (i) Draw the structural formula of compounds W, X and Yarrow_forwardWrite equations for the reaction of 2-butanol with reagent. Where you predict no reaction, write NR. Q) PCCarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


