
Concept explainers
Stanozolol is an anabolic steroid that promotes muscle growth. Although stanozolol has been used by athletes and body builders, many physical and psychological problems result from prolonged use and it is banned in competitive sports.
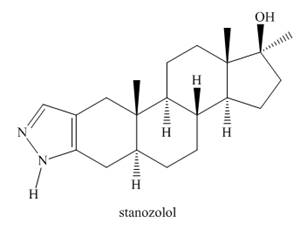
a. Explain why the nitrogen heterocycle—a pyrazole ring—is aromatic.
b. In what type of orbital is the lone pair on each
c. Draw all reasonable resonance structures for stanozolol.
d. Explain why the
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
ORGANIC CHEMISTRY-W/ACCESS >CUSTOM<
- Explain why each compound is aromatic, anti aromatic or non aromatic? Explainarrow_forwardDraw an example a) an aromatic heterocycle b) benzyne. c) an aromatic cation d) an aromatic anionarrow_forward(a) Explain why an alkylamine is more basic than ammonia?(b) How would you convert(i) Aniline to nitrobenzene (ii) Aniline to iodobenzenearrow_forward
- Draw the structure of A, an intermediate in the synthesis of the antipsychotic drug risperidone. Explain why three rings in risperidone are considered aromatic.arrow_forwardAmino acids such as glycine are the building blocks of large molecules called proteins that give structure to muscle, tendon, hair, and nails. a.) Explain why glycine does not actually exist in the form with all atomsuncharged, but actually exists as a salt called a zwitterion. b.) What product is formed when glycine is treated with concentratedHCl?c.) What product is formed when glycine is treated with NaOH?arrow_forwardWhy is an alkylamine more basic than ammononia?arrow_forward
- Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed to synthesize the drug?arrow_forwardCould you explain why the highlighted group on the right side of the double bond has a higher priority than the lower group on the right side of the double bond. I use this information to determine E/Z assigmentsarrow_forwardDraw the structure of A, an intermediate in the synthesis of theantipsychotic drug risperidone. Explain why three rings in risperidone are considered aromatic.arrow_forward
- a. Which box contains a carbocyclic unsaturation? b. Which box contains an aryl structure? c. Which box contains carboxyl group? d. Which box contains a sulfide group? e. Which box contains an amide group?arrow_forwarda.How many π electrons does C contain? b.How many π electrons are delocalized in the ring? c.Explain why C is aromatic.arrow_forwardAmino acids such as glycine are the building blocks of large molecule.called proteins that give structure to muscle, tendon, hair, and nails.a.Explain why glycine does not actually exist in the form with all atoms uncharged, but actually exists as a salt called a zwitterion. b.What product is formed when glycine is treated with concentrated HCl? c. What product is formed when glycine is treated with NaOH?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

