
Concept explainers
Interpretation:
Four examples for beta-lactam antibiotics have to be drawn. The functional group in each beta-lactam antibiotic and the common feauture in all the four examples have to be identified.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which are determine the characteristic reactions taking place in it.
Carboxylic acid: One
Ester: One
Amide: One
Alkene: Unsaturated hydrocarbons having atleast one double bond between two carbon atoms are known as alkenes.
Ether: Ether is a group of organic compound where two aryl or alkyl groups are connected by an oxygen atom. It is represented as
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
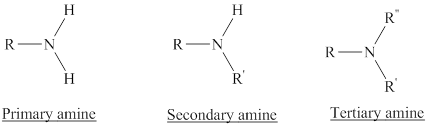
Lactam: They are cyclic amides where the carbonyl carbon part of carboxylic acid group and nitrogen part of amine group of the same compound attached forming a ring. Beta-lactam is a membered ring with two carbon atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
FUND.OF GEN CHEM CHAP 1-13 W/ACCESS
- Why is the structure of ornithine so similar to the structure of citrulline?arrow_forwardWhy Ranitidine is better than Cemitidine? Please explain at your own words.arrow_forwardWhat product is formed when a solution of A and B is treated with mild base? This reaction is the first step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forward
- Draw the structure of the PTH derivative product you would obtain by Edman degradation of the following peptides: (a) I-L-P-F (b) D-T-S-G-Aarrow_forwardName the heterocyclic substituent found in the molecule shown according to the general type of nitrogen-containing ring to which it belongs.arrow_forwardWhy 14C@phenylalanine is used ?arrow_forward
- Draw structures of the following compounds:(a) cis-Hept-3-ene(b) cis-4-Methylpent-2-ene(c) trans-2,5-Dimethylhex-3-enearrow_forwardHow do the functional groups of rilpivirine add to its function in the human body? How does rilpivirine travel through the body?arrow_forwardHow can chirality and stereoisomers influence the pharmacology, bioactivity, toxicology, pharmacokinetics, and metabolism of ibuprofen? Please provide a detailed summary of what might happen if, for instance, the R enantiomer isn't able to be inverted to the bioactive S enantiomer.arrow_forward
- Draw structures corresponding to the following names:(a) 3-Methylhexan-1-ol (b) 1-Methyl-3-propylcyclopentanol(c) 2,2-Dimethylhexan-3-ol (d) Heptan-3-ol(e) 2,3-Diethylcyclohexanolarrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. a) d- b) S- c) R- d) I-arrow_forwardWhat is the structural difference between a hemiacetal and an acetal?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





