
(a)
Interpretation: The IUPAC name for Megatomoic acid has to be given.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
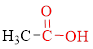
Nomenclature of
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the
alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
E-Z designators are used as like cis-trans terminology for non-similar groups attached
In E-Z designations, the groups attached to vinylic positions are checked by their priority on the basis of higher molecular weight. If the higher priority groups are on the same sides, then the configuration is designated as Z. If the higher priority groups are on the opposite sides, then the configuration is designated as E.
(b)
Interpretation: The number of possible stereoisomers for the given compound has to be stated.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
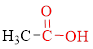
The stereoisomerism is the arrangement of atoms in molecules whose connectivity remains the same but their arrangement in different in each isomer.
The two molecules are described as stereoisomers if they are made of the same atoms connected in the same sequence, but the atoms are positions differently in space
E-Z designators are used as like cis-trans terminology for non-similar groups attached alkenes.
In E-Z designations, the groups attached to vinylic positions are checked by their priority on the basis of higher molecular weight. If the higher priority groups are on the same sides, then the configuration is designated as Z. If the higher priority groups are on the opposite sides, then the configuration is designated as E.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry
- Provide the proper systematic IUPAC name for the following compounds. Please assign E, Z, R, and S.arrow_forwardGive IUPAC names for the following compounds:arrow_forwardExplain why tetrahydrofuran has a higher boiling point and is much more water soluble than furan, even though both compounds are cyclic ethers containing four carbons.arrow_forward
- Draw and name all constitutionally isomeric alcohols with the molecular formula C4H10Oarrow_forwardCompound A and compound B are different alcohols with the same molecular formula of C4H10O. When reacted with chromic acid (H2CrO4), compound A and compound B produce compound C and compound D, respectively. Compound C has a molecular formula of C4H8O2. Compound E is obtained when compound A reacts with pyridinium chlorochromate (PCC) in a solvent such as dichloromethane (CH2Cl2). Draw the structure of compound A, B, C, D and E.arrow_forwardGive the IUPAC name for each compound (D, E, F)arrow_forward
- Provide the IUPAC name of the compound shown below? Please don't forget to include the stereocenters in the name.arrow_forward2,3-Dimethylfumaric acid has a molecular formula C6H8O4. It undergoes oxidativecleavage to form two identical compound N. Compound N is then reacted withethylmagnesium bromide to form compound O. Compound O is then hydrolysed inacidic condition to form compound P. Draw the structure of compound N, O and P. PLEASE PROVIDE CLEAR HANDWRITING, and explantionarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

