
Concept explainers
Interpretation:
The dicarboxylic acid and diamine which are needed to prepare the following polyamide should be determined:
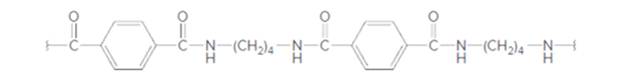
Concept Introduction:
On the basis of type of polymers, polymerization reaction can be two types; addition polymerization and condensation polymerization. The addition polymerization occurs between monomer units with unsaturated bonds between carbon atoms. For example; polymerization of
Condensation polymerization occurs with those monomers which have some
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Write the structures of the following carboxylic acids and esters: a) Salicylic acid b) Benzoic acid c) tert-butyl butanoate d) isopropyl benzoatearrow_forwardBenzoic acid reacts with ethylamine to form what kind of amidearrow_forwardDraw the structure if the following compounds. benzoic propanoic anhyydride octyl butanoate 30methylhexanoyl chloridearrow_forward
- What is the product of pentene and water under acidic conditions? Group of answer choices pentanol 2-pentanone pentanoic acid pentanalarrow_forwardHow could butanone be prepared from butane?arrow_forwardWhat type of chemical reaction does carboxylic acid undergo in the following situations? 1. synthesis of methyl salicylate 2. reaction with strong alkali 3. occurs in aqueous solution 4. release of hydrogen gasarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,


