
Concept explainers
(a)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(a)
Answer to Problem 18.109EP
The name of the given structure is methyl-
Explanation of Solution
The Fisher projection formula for D-allose is,
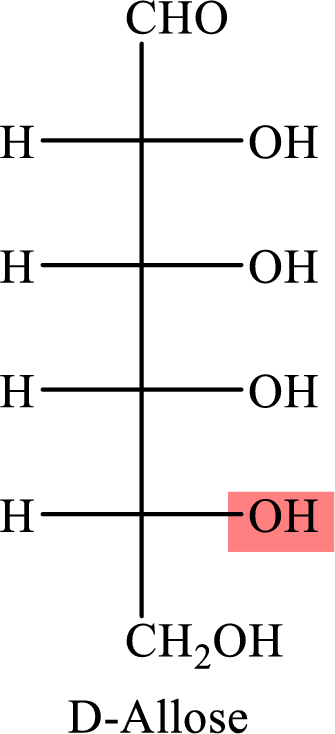
The structure given in Problem 18-103 is,
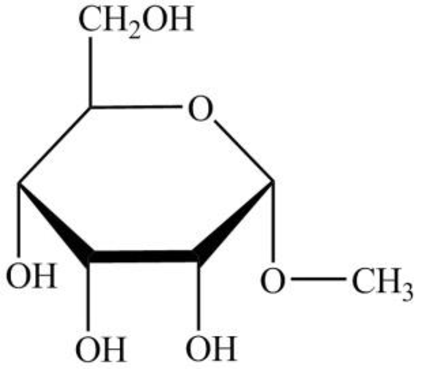
The above structure is the cyclic form of monosaccharide D-allose. Both the
(b)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(b)
Answer to Problem 18.109EP
The name of the given structure is ethyl-
Explanation of Solution
The Fisher projection formula for D-altrose is,
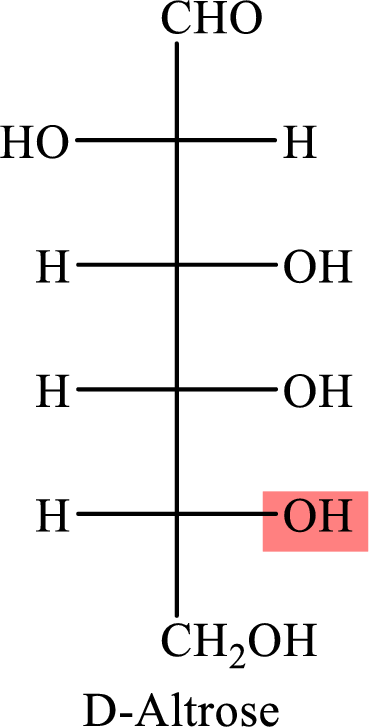
The structure given in Problem 18-103 is,
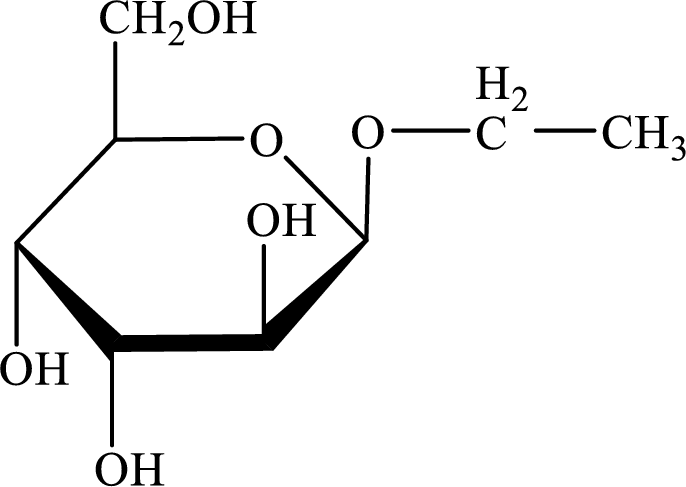
The above structure is the cyclic form of monosaccharide D- altrose. Both the
(c)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(c)
Answer to Problem 18.109EP
The name of the given structure is ethyl-
Explanation of Solution
The Fisher projection formula for D-fructose is,
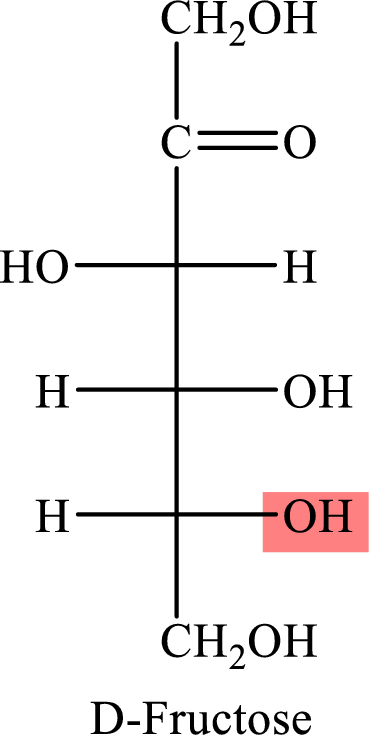
The structure given in Problem 18-103 is,
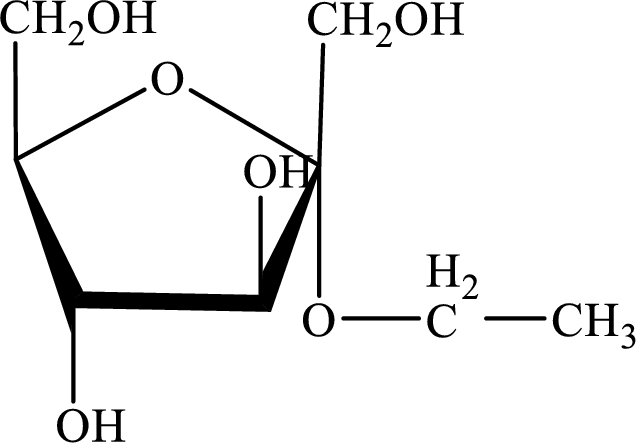
The above structure is the cyclic form of monosaccharide D-fructose. Both the
(d)
Interpretation: The name of the given compound has to be stated.
Concept introduction: In a cyclic monosaccharide, the replacement of an
(d)
Answer to Problem 18.109EP
The name of the given structure is methyl-
Explanation of Solution
The Fisher projection formula for D-glucose is,
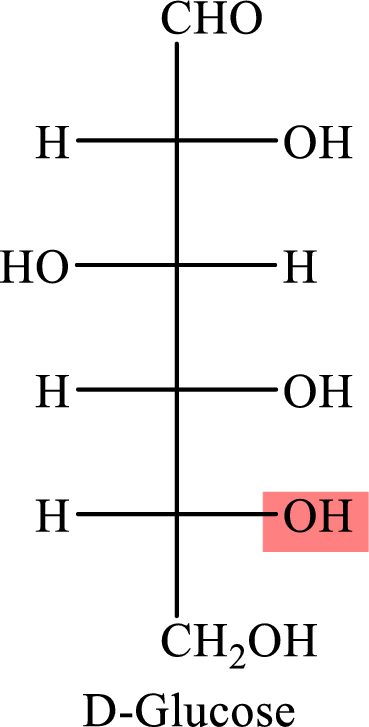
The structure given in Problem 18-103 is,
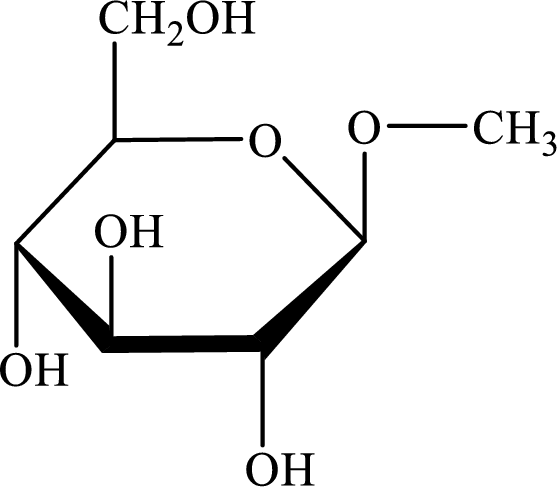
The above structure is the cyclic form of monosaccharide D-glucose. Both the
Want to see more full solutions like this?
Chapter 18 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- 1 Hyaluronic acid acts as a lubricant in the synovial fluid of joints. In rheumatoid arthritis, inflammation breaks hyaluronic acid down to smaller molecules. Under these conditions, what happens to the lubricating power of the synovial fluid?arrow_forwardC) Complete Oxidation or Burning of Organic Compounds 5) Propane, C,Hg, burns in air. 6) Acetone (nail polish remover), CH3COCH3, is completely oxidized.arrow_forwardWhich of the following statements is not true for vital force theory? O Life force was necessary to produce organic compounds Organic compounds can only be produced by and within living things O Inorganic materials did not contain the "vital force" of life None of the abovearrow_forward
- Which of the following is a fatty acid which is a solid at room temperature? OH H. OH OHarrow_forwardWhat is the balanced chemical equation when gaseous butanol, C4H9OH, undergoes combustion?arrow_forwardName the organic reactant required to react with ammonia to produce hexan-1-amine and HBr.arrow_forward
- Aspirin has the following structure, what are the names of the functional groups present? OH CH3 amide alcohol ether ester ketone aldehyde phenol carboxylic acidarrow_forwardIf an oxidation reaction occurs with an alcohol, what are you removing and what are you adding to your molecule?arrow_forward4 What type of organic molecule is this? ОН ketone alcohol aldehyde organic acidarrow_forward
- The functional group present in proteins is aldehyde a b amide hydroxyl C d. ketonearrow_forwardIndicate which classes of organic compounds are present in the following molecule. H₂Narrow_forwardWhat functional groups are present in a carbohydrate molecule? a. Carboxyl and carbonyl groups b. Alcohol and carboxyl groups c. Hydroxyl and carbonyl groups d. Hydroxyl and hydrogen groupsarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





